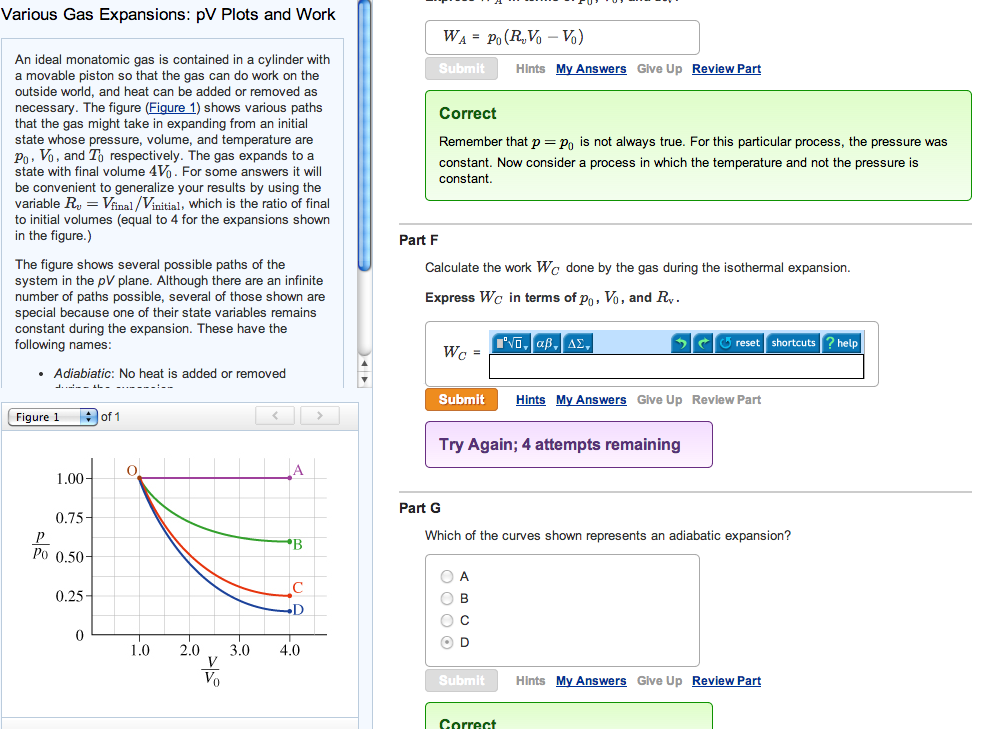
Various Gas Expansions: pV Plots and Work An ideal monatomic gas is contained in a cylinder...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Various Gas Expansions: pV Plots and Work An ideal monatomic gas is contained in a cylinder with a movable piston so that the gas can do work on the outside world, and heat can be added or removed as necessary. The figure (Figure 1) shows various paths that the gas might take in expanding from an initial state whose pressure, volume, and temperature are Vo, and To respectively. The gas expands to a state with final volume 4V₁. For some answers it will be convenient to generalize your results by using the variable R₂ = Vfinal/Vinitial, which is the ratio of final to initial volumes (equal to 4 for the expansions shown in the figure.) The figure shows several possible paths of the system in the pV plane. Although there are an infinite number of paths possible, several of those shown are special because one of their state variables remains constant during the expansion. These have the following names: • Adiabiatic: No heat is added or removed Figure 1 1.00- 0.75- Po 0.50- 0.25- 0 - of 1 O 1.0 2.0 D 3.0 4.0 WA PO (R₂Vo - Vo) Submit Correct Remember that p = Po is not always true. For this particular process, the pressure was constant. Now consider a process in which the temperature and not the pressure is constant. Part F Calculate the work We done by the gas during the isothermal expansion. Express Wc in terms of po, Vo, and R. ΓΕ, αβ, ΔΣ, 5 reset shortcuts ? help Wc Submit Hints My Answers Give Up Review Part Try Again; 4 attempts remaining O A Ов O C OD Part G Which of the curves shown represents an adiabatic expansion? Submit Hints My Answers Give Up Review Part Correct Hints My Answers Give Up Review Part Various Gas Expansions: pV Plots and Work An ideal monatomic gas is contained in a cylinder with a movable piston so that the gas can do work on the outside world, and heat can be added or removed as necessary. The figure (Figure 1) shows various paths that the gas might take in expanding from an initial state whose pressure, volume, and temperature are Vo, and To respectively. The gas expands to a state with final volume 4V₁. For some answers it will be convenient to generalize your results by using the variable R₂ = Vfinal/Vinitial, which is the ratio of final to initial volumes (equal to 4 for the expansions shown in the figure.) The figure shows several possible paths of the system in the pV plane. Although there are an infinite number of paths possible, several of those shown are special because one of their state variables remains constant during the expansion. These have the following names: • Adiabiatic: No heat is added or removed Figure 1 1.00- 0.75- Po 0.50- 0.25- 0 - of 1 O 1.0 2.0 D 3.0 4.0 WA PO (R₂Vo - Vo) Submit Correct Remember that p = Po is not always true. For this particular process, the pressure was constant. Now consider a process in which the temperature and not the pressure is constant. Part F Calculate the work We done by the gas during the isothermal expansion. Express Wc in terms of po, Vo, and R. ΓΕ, αβ, ΔΣ, 5 reset shortcuts ? help Wc Submit Hints My Answers Give Up Review Part Try Again; 4 attempts remaining O A Ов O C OD Part G Which of the curves shown represents an adiabatic expansion? Submit Hints My Answers Give Up Review Part Correct Hints My Answers Give Up Review Part
Expert Answer:
Answer rating: 100% (QA)
Solution Part F Work done in isothermal process is given by W c nRT ln V f ... View the full answer

Related Book For 

Posted Date:
Students also viewed these physics questions
-
Please show separate graphs for each part to receive appropriate credit for each part. Feel free to use Powerpoint, Excel, or any software that can do the job, OR neatly draw by hand. If drawing by...
-
please show me er diagram as well as explain it between room and customer there will be many to many cardinality ratio please convert it into one to many .also show the entity hotel have staff and...
-
3. Spicy City (SC) and Village Kitchen (VK) are the only two Chinese restaurants serv- ing Mapo Tofu in Clairemont Mesa, San Diego. They use very similar methods to cook their Mapo Tofu, so they have...
-
Uniform rod AB of length l and mass m lies in a vertical plane and is acted upon by a couple M. The ends of the rod are connected to small rollers which rest against frictionless surfaces. (a)...
-
Sherrye Cravens owns Cravenss Consulting Service. At the beginning of September, her firms financial records showed the following assets, liabilities, and owners equity. INSTRUCTIONS Set up an...
-
Determine the accounts payable to revenues ratio for the construction company in Figures 6-1 and 6-2. What insight does this give you into the companys financial operations? Figures 6-1 WEST MOUNTAIN...
-
Refer to the information in Problem 21-1B. Tohono Companys actual income statement for 2017 follows. Required 1. Prepare a flexible budget performance report for 2017. Analysis Component 2. Analyze...
-
On March 20, Dodys petty cash fund of $100 is replenished when the fund contains $9 in cash and receipts for postage $52, freight-out $26, and travel expense $10. Give the journal entry to record the...
-
15. Jenny borrows $20,000 for her car at an interest rate of 2.5% to be paid off over five years, during which time the inflation rate averages 7%
-
Calculate Gillian Pool and Spa Supplies average annual compound growth rate of sales and analyze its earnings performance for the past 5 years.
-
A Lottery winner chooses to receive a yearly distribution of her winnings instead of a lump sum payout. She will receive a payment of 8,972,945 every year starting today for the next 20 years. Assume...
-
Where would a gun buyback program fall under? prepare the budget prepares the council agenda may have veto on council actions appoints department heads supervises the city bureaucracy approves the...
-
You borrow $ 540,000 to buy a house worth $600,000. The interest rate is 2.4 % pa compounding monthly and the fully amortising loan term is 30 years. A loan repayment schedule is shown below, with...
-
How does the molecular friction near the surface affect the flow in the planetary boundary layer? List different types of energy fluxes at an ideal surface, write the surface energy balance equation,...
-
Three companies are selling bonds with the following cash flows. $1,000 at the end of year 5 $100 per year for five years. How much would each of these bonds sell for if the companies credit rating...
-
Supposing that all of the remaining 300 shares were liquidated in December 200B at P100 per share because of bankruptcy of X Corporation. What would be the capital loss in December 200B? Explain...
-
What is the marketing mix, also known as the 4Ps?
-
The Thomas Corporation was organized on Jan. 1, 2020. On Dec. 31, 2021, the corporation lost most of its inventory in a warehouse fire before the year-end count of inventory was to take place. just...
-
A mirror at an amusement park shows an upright image of any person who stands 1.9 m in front of it. If the image is three times the person's height, what is the radius of curvature of the mirror?...
-
A 125-kg astronaut (including space suit) acquires a speed of 2.50 m/s by pushing off with her legs from a 1900-kg space capsule. (a) What is the change in speed of the space capsule? (b) If the push...
-
How far apart an object and an image are formed by an 85-cm-focal-length converging lens if the image is 3.25 x larger than the object and is real?
-
In a large office building, there is a woman dead in her locked office, sitting at her desk. Except for the desk, chair, and contents on the desk, the office is empty and the air conditioner is...
-
How many birthdays does the average person have?
-
A murderer is condemned to death. He has to choose between three rooms. The first is full of raging fires, the second is full of assassins with loaded guns, and the third is full of lions that havent...

Study smarter with the SolutionInn App


