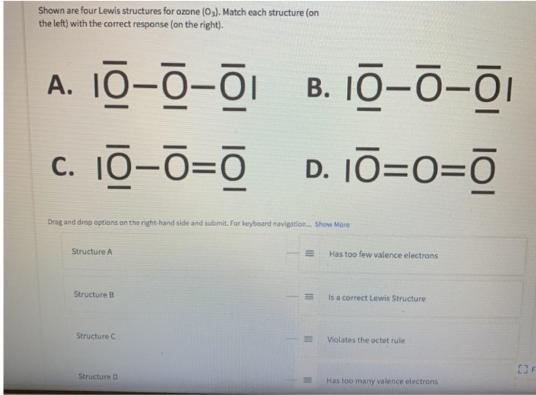
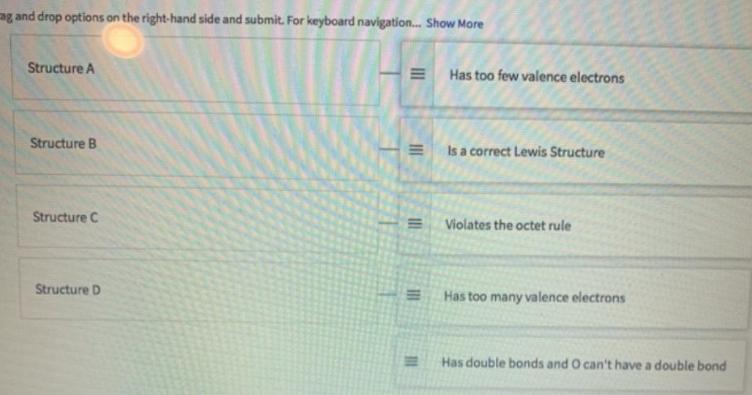
Shown are four Lewis structures for ozone (0,). Match each structure (on the left) with the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Shown are four Lewis structures for ozone (0,). Match each structure (on the left) with the correct response (on the right). A. 10-0-01 B. 10-ō-ōi c. 10-0=0 D. 10=0=O Drag and dine optians an the rght hand side and mit. For beybeard navigation tho Mare Structure A Has too few valence electrans Structure is a correct Lewis Structure Structure C Violates the uctet rule Structurm D Has too many valence electrons g and drop options on the right-hand side and submit. For keyboard navigation. Show More Structure A Has too few valence electrons Structure B Is a correct Lewis Structure Structure C Violates the octet rule Structure D Has too many valence electrons Has double bonds and O can't have a double bond II II Shown are four Lewis structures for ozone (0,). Match each structure (on the left) with the correct response (on the right). A. 10-0-01 B. 10-ō-ōi c. 10-0=0 D. 10=0=O Drag and dine optians an the rght hand side and mit. For beybeard navigation tho Mare Structure A Has too few valence electrans Structure is a correct Lewis Structure Structure C Violates the uctet rule Structurm D Has too many valence electrons g and drop options on the right-hand side and submit. For keyboard navigation. Show More Structure A Has too few valence electrons Structure B Is a correct Lewis Structure Structure C Violates the octet rule Structure D Has too many valence electrons Has double bonds and O can't have a double bond II II
Expert Answer:
Answer rating: 100% (QA)
In O 3 total number of valence electrons 63 18 es In structure A total num... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
6.36 For the following mechanism, identify the sequence of arrow-pushing patterns: :H :A R :R -R H H 4,0 R R R
-
+/ F(+) F'(+)=-1-in(f(+); f(+)=? Show your all work
-
1. Which of the following is not normally a tax-paying entity under the Federal income tax? a. The regular corporation b. The estate of a deceased individual c. The partnership entity d. A trust for...
-
Prove that the function x2 x - 1 | f (x) = x 1)(x 2) is differentiable for all r E (-0, 1) U (1, 2) U (2, ). - |
-
Estimate the enthalpy of reaction for the equilibrium reaction CH4 + 2O2 CO2 + 2H2O at 2500 K, using (a) enthalpy data and (b) KP data. Obtain enthalpy and entropy properties from EES.
-
The balances of certain accounts of Camille Corporation on April 30, 20X1, were as follows: Sales $ 330,000 Sales Returns and Allowances 4,500 The firms net sales are subject to an 8 percent sales...
-
Repeat Problem 8.106 if the downspout is circular. Problem 8.106 Rainwater flows through the galvanized iron downspout shown in Fig. P8.106 at a rate of \(0.006 \mathrm{~m}^{3} / \mathrm{s}\)....
-
Frate Company was formed on January 1, 2014. The following information is available from Frates inventory records for Product Ply: .:. A physical inventory on December 31, 2014, shows 1,600 units on...
-
Arvin, Inc., produces two products, ins and outs, in a single process. The joint costs of this process were $77,300, and 14,000 units of ins and 36,000 units of outs were produced. Separable...
-
Suppose that Kate and Anne enter into a pooling arrangement. Assume that both women have the following loss distributions and that losses are independent. $50,000 with probability of 0.005 $20,000...
-
When preforming credit risk analysis, try to assess the following: Expected Credit Loss= Chance of Default Loss Given Default Comment on overall assessment of the company's credit risk and chance of...
-
Lett Incorporateds stock price is now $50, but it is expected either to rise by a factor of 1.5 or to fall by a factor of 0.7 by the end of the year. There is a call option on Letts stock with a...
-
Bob Keeley wants to buy a new high-end audio system for his car. The system is being sold by two dealers in town, both of whom sell the equipment for the same price of $2,000. Bob can buy the...
-
The discussion in the chapter focuses on the square-well fluid, but the same reasoning is equally applicable for any model potential function. Illustrate your grasp of this reasoning with some...
-
At age 19, Linda Sayers is in the middle of her second year of studies at a community college in Phoenix. She has done well in her course work; majoring in prebusiness studies, she currently has a...
-
Morris Corporation has the following information on its balance sheets: Cash = $40, accounts receivable = $30, inventories= $100, net fixed assets 5 $500, accounts payable = $20, accruals= $10,...
-
When the Centers for Medicare and Medicaid Services (CMS) makes a payment using the Prospective Payment System, what is the Medicare payment based on?
-
7. Baladna wants to analyze process that includes delivery by suppliers, production inside the company, transportation to to its customers and information systems. Then it also wants to find out...
-
A photocell is a device used to measure the intensity of light. In a certain experiment, when light of wavelength 630 nm is directed onto the photocell, electrons are emitted at the rate of 2.6 x...
-
(a)Which electrode of a voltaic cell, the cathode or the anode, corresponds to the higher potential energy for the electrons? (b) What are the units for electrical potential? How does this unit...
-
You have studied the gas-phase oxidation of HBr by O2: 4 HBr(g) + O2(g) 2 H2O(g) + 2 Br2(g) You find the reaction to be first order with respect to HBr and first order with respect to O2. You...
-
Alma, Inc., manufactures high-pressure cleaners for large transportation companies. It uses standard costing and allocates variable and fixed manufacturing overhead based on machine-hours. For each...
-
Michael Roberts is a cost accountant and business analyst for Darby Design Company (DDC), which manufactures expensive brass doorknobs. DDC uses two direct-cost categories: direct materials and...
-
Omegas Fleet Feet, Inc., produces dance shoes for stores all over the world. While the pairs of shoes are boxed individually, they are crated and shipped in batches. The shipping department records...

Study smarter with the SolutionInn App


