7.36. Sketch a temperature-composition phase diagram (see i Figures 7.14 and 7.15 for examples) for the...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
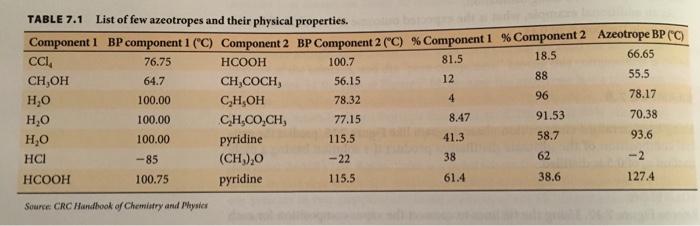
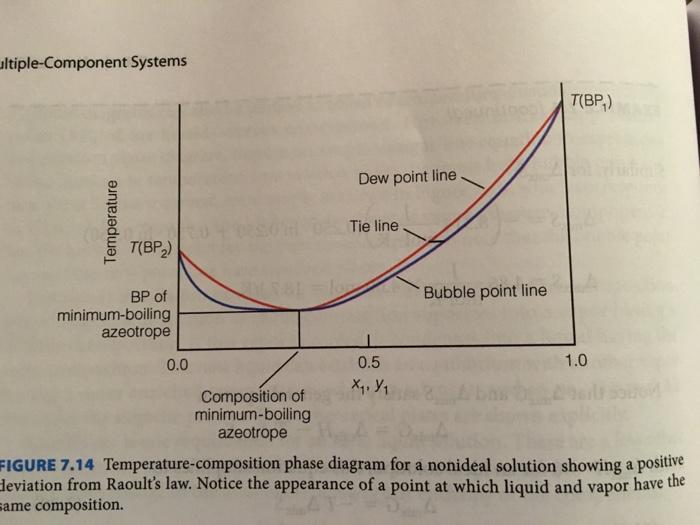
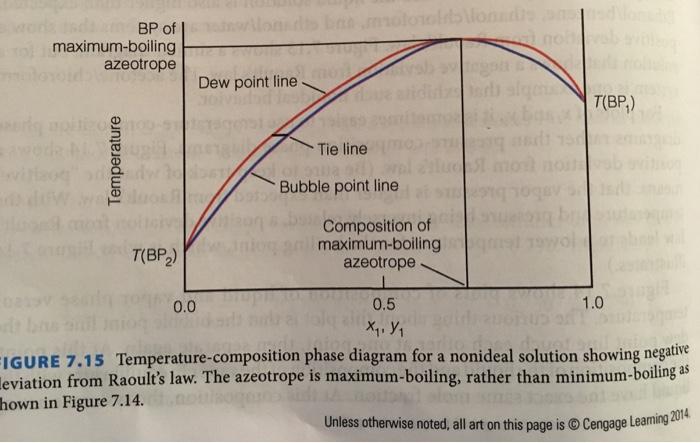
7.36. Sketch a temperature-composition phase diagram (see i Figures 7.14 and 7.15 for examples) for the CCl/HCOOH and HCOOH/pyridine solutions, using data from Table 7.1. How are the two phase diagrams different? How are they similar? TABLE 7.1 List of few azeotropes and their physical properties. Component 1 BP component 1 ("C) Component 2 BP Component 2 ("C) % Component 1 % Component 2 Azeotrope BP ("C) CC, 81.5 18.5 66.65 76.75 HCOOH 100.7 CH,OH 12 88 55,5 64.7 CH,COCH, 56.15 H,0 4 96 78.17 100.00 C,H,OH 78.32 100.00 CH,CO,CH, 8.47 91.53 70.38 77.15 H,O pyridine 115.5 41.3 58.7 93.6 100.00 HCI -85 (CH,),0 -22 38 62 -2 НСООН 100.75 pyridine 115.5 61.4 38.6 127.4 Source: CRC Handbook of Chemistry and Physies ultiple-Component Systems T(BP,) MAI Dew point line Tie line T(BP,) Bubble point line BP of minimum-boiling azeotrope 0.0 0.5 1.0 Composition of minimum-boiling azeotrope FIGURE 7.14 Temperature-composition phase diagram for a nonideal solution showing a deviation from Raoult's law. Notice the appearance of a point at which liquid and vapor have the same composition. positive Temperature Unless otherwise noted, all art on this page is © Cengage Leaming 2014 wodk sdam BP of | ilonsdts bammoloroltslonads dlon ol maximum-boiling noal azeotrope Dew point line adh alge T(BP,) Tie line lust pente Bubble point line br Composition of maximum-boiling azeotrope T(BP,) 0.0 0.5 1.0 X, Y, FIGURE 7.15 Temperature-composition phase diagram for a nonideal solution showing negative leviation from Raoult's law. The azeotrope is maximum-boiling, rather than minimum-boiling as hown in Figure 7.14. Temperature 7.36. Sketch a temperature-composition phase diagram (see i Figures 7.14 and 7.15 for examples) for the CCl/HCOOH and HCOOH/pyridine solutions, using data from Table 7.1. How are the two phase diagrams different? How are they similar? TABLE 7.1 List of few azeotropes and their physical properties. Component 1 BP component 1 ("C) Component 2 BP Component 2 ("C) % Component 1 % Component 2 Azeotrope BP ("C) CC, 81.5 18.5 66.65 76.75 HCOOH 100.7 CH,OH 12 88 55,5 64.7 CH,COCH, 56.15 H,0 4 96 78.17 100.00 C,H,OH 78.32 100.00 CH,CO,CH, 8.47 91.53 70.38 77.15 H,O pyridine 115.5 41.3 58.7 93.6 100.00 HCI -85 (CH,),0 -22 38 62 -2 НСООН 100.75 pyridine 115.5 61.4 38.6 127.4 Source: CRC Handbook of Chemistry and Physies ultiple-Component Systems T(BP,) MAI Dew point line Tie line T(BP,) Bubble point line BP of minimum-boiling azeotrope 0.0 0.5 1.0 Composition of minimum-boiling azeotrope FIGURE 7.14 Temperature-composition phase diagram for a nonideal solution showing a deviation from Raoult's law. Notice the appearance of a point at which liquid and vapor have the same composition. positive Temperature Unless otherwise noted, all art on this page is © Cengage Leaming 2014 wodk sdam BP of | ilonsdts bammoloroltslonads dlon ol maximum-boiling noal azeotrope Dew point line adh alge T(BP,) Tie line lust pente Bubble point line br Composition of maximum-boiling azeotrope T(BP,) 0.0 0.5 1.0 X, Y, FIGURE 7.15 Temperature-composition phase diagram for a nonideal solution showing negative leviation from Raoult's law. The azeotrope is maximum-boiling, rather than minimum-boiling as hown in Figure 7.14. Temperature
Expert Answer:
Answer rating: 100% (QA)
From the table we can see that CCl4HCOOH solution froms a minimum boiling azeotropes Hence it shows ... View the full answer

Related Book For 

Fundamentals of Investment Management
ISBN: 978-0078034626
10th edition
Authors: Geoffrey Hirt, Stanley Block
Posted Date:
Students also viewed these physics questions
-
Using data from Table 165 on page 426, assume you purchase a December 1100 (strike price) S&P 500 put option. Compute your total dollar profit or loss if the index has the following values at...
-
Using data from Table 8.4 on bond enthalpies, show that the more C---H bonds a molecule has compared to C-----O and O----H bonds, the more energy it can store.
-
Using data from Table 2.1 (p. 34), illustrate on the following graphs real GDP and population growth since 2000 (in the manner of Figure 2.1) for the nations indicated. China Canada Zimbabwe 100 100...
-
Given the project network that follows, compute the early, late, and slack times for the project. Be sure to show the early finish and late start times on yournetwork. 20 15 25 10 10 Slack IS B
-
Create the following matrix by typing one command. Do not type individual elements explicitly. E=100000 00 5 4 3 0021
-
Consider the following premerger information about a bidding firm (Firm B) and a target firm (Firm T). Assume that both firms have no debt outstanding. Firm B Firm T Shares...
-
The van der Waals equation of state is given by (a) \(\left(P+\frac{a}{V} ight)(V-b)=R T\) (b) \(\left(P+\frac{a}{V^{2}} ight)(V-b)=R T\) (c) \(\left(P+\frac{a}{V^{2}} ight)(V+b)=R T\) (d) None of...
-
A vapor mixture containing 30 mole% benzene and 70% toluene at 1 atm is cooled isobarically j, a closed container from an initial temperature of 115C. Use the Txy diagram of Figure 6.4-1 to answer...
-
What type of capacity planning tools would you use to ensure that your IT workplace is operating at optimal efficiency?
-
In Intuit QuickBooks how do I record this transaction?... Wrote check number 10010 to City Credit Union for payment of amount due (City Credit Union in the Account/Category field); $110.35.
-
Discuss the answers with your tutor. 1. Discuss the disclosure issues addressed by the following: (a) The Securities Act 1933 (b) The Security Exchange Act 1934 (c) The Foreign Corruption Practices...
-
A business sold one of their trucks at the end of 2019. The truck originally cost $80,500, had been in use for 4 years, and had accumulated depreciation of $46,000 at the time of sale. The sale of...
-
Activity Rates and Activity-Based Product Costing Hammer Company produces a variety of electronic equipment. One of its plants produces two laser printers: the deluxe and the regular. At the...
-
Explain the necessary components of an executive summary and share the city and only one recommendation that you have proposed to expand and grow by building a new hospital. Describe two components...
-
Does the hedonic treadmill help or hinder the pursuit of happiness? You must explore both the pros and cons here. From or class material, or any other source, give reasons why?
-
1. As explained in chapter one, accounting is a measurement and communication discipline based on rules referred to as generally accepted accounting (GAAP). Explain what are pros and cons of GAAP? ...
-
Cam corp. is a manufacturer of wholesale goods. It currently sells to two products. Product A priced at 2,000 and B priced at 1,000. Cam Corp is trying to understand the true profit for each product...
-
What are the three kinds of research types? Explain each type.
-
Compute the duration for the data in problem 1. Use an approach similar to that in Table 182 on page 469. A discount rate of 13 percent should be applied.
-
Explain the difference between a cash account and a margin account.
-
You have invested $1,000 in a 13 percent coupon bond that matures in five years. This bond is held in your individual retirement account, and you are not concerned about tax consequences. You are...
-
The finite difference method requires the use of finite difference approximations in a. governing differential equation only b. boundary conditions only c. governing differential equation as well as...
-
Fill in the Blanks. In a conditionally stable method, the use of \(\Delta t\) larger than \(\Delta t_{\text {cri }}\) makes the method _____________.
-
True or False. For a beam with grid points \(-1,1,2,3, \ldots\), the central difference approximation of a simply supported end condition at grid point 1 is given by \(W_{-1}=W_{2}\).

Study smarter with the SolutionInn App


