Sodium nitrite, NaNO, has a molar mass of 85.0 g/mol. What volume of 1.60 M NaNO;...
Fantastic news! We've Found the answer you've been seeking!
Question:
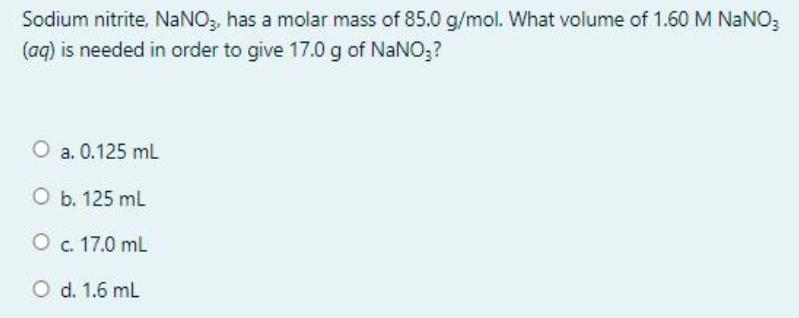
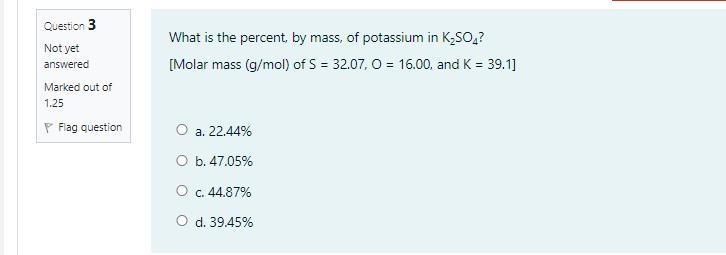

Transcribed Image Text:
Sodium nitrite, NaNO, has a molar mass of 85.0 g/mol. What volume of 1.60 M NaNO; (aq) is needed in order to give 17.0 g of NaNO;? O a. 0.125 mL O b. 125 mL O c. 17.0 ml O d. 1.6 mL Question 3 What is the percent, by mass, of potassium in K;SO,? Not yet [Molar mass (g/mol) of S = 32.07, O = 16.00, and K = 39.1] answered Marked out of 1.25 P Flag question O a. 22.44% а. O b. 47.05% O c. 44.87% O d. 39.45% Question 2 Given the thermochemical equation: 250,(g) + O2(g) – 2 so;(g) AH ren = -198 kJ How much heat is evolved when 192 g of SOz is burned? [molar mass (g/mol) of S = 32.00, and O = 16.00] Not yet answered Marked out of 1.25 P Flag question О а. 495 k O b. 297 kJ О с. 198 k O d. 396 kJ Sodium nitrite, NaNO, has a molar mass of 85.0 g/mol. What volume of 1.60 M NaNO; (aq) is needed in order to give 17.0 g of NaNO;? O a. 0.125 mL O b. 125 mL O c. 17.0 ml O d. 1.6 mL Question 3 What is the percent, by mass, of potassium in K;SO,? Not yet [Molar mass (g/mol) of S = 32.07, O = 16.00, and K = 39.1] answered Marked out of 1.25 P Flag question O a. 22.44% а. O b. 47.05% O c. 44.87% O d. 39.45% Question 2 Given the thermochemical equation: 250,(g) + O2(g) – 2 so;(g) AH ren = -198 kJ How much heat is evolved when 192 g of SOz is burned? [molar mass (g/mol) of S = 32.00, and O = 16.00] Not yet answered Marked out of 1.25 P Flag question О а. 495 k O b. 297 kJ О с. 198 k O d. 396 kJ
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A certain material has a molar mass of 20.0 g/mol, a Fermi energy of 5.00eV, and 2 valence electrons per atom. What is the density (g/cm3)?
-
A starch has a molar mass of 3.20 1004 g/mol. If 0.759 g of this starch is dissolved in 112 mL of solution, what is the osmotic pressure, in torr, at 25.00C?
-
(a) Oxygen (02) has a molar mass of 32.0 g/mol what is the average translational kinetic energy of an oxygen molecule at a temperature of 300 K? (b) What is the average value of the square of its...
-
Researchers examined forecasters' interest rate predictions for 34 quarters to see whether the predictions corresponded to what actually happened. The 2 x 2 contingency table below shows the...
-
The mean of the waiting times in an emergency room is 80.2 minutes with a standard deviation of 10.5 minutes for people who are admitted for additional treatment. The mean waiting time for patients...
-
Max Industries Ltd. borrowed money by issuing a \(\$ 10,000\) 6.5\%, 10-year bond. Assume the issue price was 94 on July \(1,2020\). 1. How much cash did Max Industries receive when it issued the...
-
The following is the adjusted trial balance of Qwik Care Clinic, Inc., for December 31,2010. Requirement 1. Journalize the closing entries at December 31. Qwik Care Clinic, Inc. Adjusted Trial...
-
Was the confiscation of all cigar manufacturers by the Cuban government in the early 1960s and expropriation or a nationalization? Was this confiscation commercial activity in which a private...
-
Metlock Corporation had 291,000 shares of common stock outstanding on January 1, 2025. On May 1, Metlock issued 29,400 shares. (a) Compute the weighted-average number of shares outstanding if the...
-
1. You borrow $50,000 for 5 years. This is an amortized load, meaning that payments are the same each month, and the loan is fully paid off with final payment. The quoted interest rate (or APR) is...
-
JCDecaux's CFO tells you the company wants to make an investment in a fast-growing and highly profitable counter-cyclical industry to smooth the firm's cash flows. What advice would you give the CFO...
-
What is the difference between a closed society and an open society? Is the United States a closed or an open society? What about Japan, the historic Soviet Union, and Great Britain?
-
Suppose Techoland and Cornsylvania form a currency union and adopt the electrocarrot as their common currency. Now suppose again that there is an increase in demand for electronic goods in both...
-
Why is H. erectus the first hominin to move out of the African continent and colonize the Old World?
-
In the face of a financial crisis such as that created by the dot.com collapse and the 9/11 terrorist attacks in the US, why would a central bank have responded by lowering interest rates and...
-
How has industrialization affected the family, kinship, marriage, and divorce?
-
Consider the following Product Structure Tree/Diagram for a stool: Stool Seat Ass'y (1) Nuts (3) Top tipped leg (1) Bottom Seat Nuts Top (1) (2) tipped leg (1) leg (1) Leg tips (2) How many Nuts...
-
The Dow Jones Industrial Average reached a high of $ 7801.63 on December 29, 1997. Recall from Example 18.4 that it reached a high of $ 1003 on November 14, 1972. The Consumer Price Index for...
-
Natural gas consists primarily of methane, CH4. It is used in a process called steam reforming to prepare a gaseous mixture of carbon monoxide and hydrogen for industrial use. CH4(g) + H2O(g) CO(g)...
-
Which of the following particles has the longest wavelength? a. An electron traveling at x meters per second b. A proton traveling at x meters per second c. A proton traveling at 2x meters per second
-
Give the names of the following ions. a. Mn2+ b. Ni2+ c. Co2+ d. Co3+
-
Give examples of non-current (or fixed) assets. In what circumstances would some of the items you have listed be current assets?
-
What is the difference between a non-current asset and a current asset?
-
You are given the following simplified statement of financial position of the Sandin Castle Company: a Calculate the current ratio. b Calculate the liquidity ratio. c After preparing the statement of...

Study smarter with the SolutionInn App


