solution is formed when the solute uniformly disperses oughout (or dissolves in) the solvent. The process...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
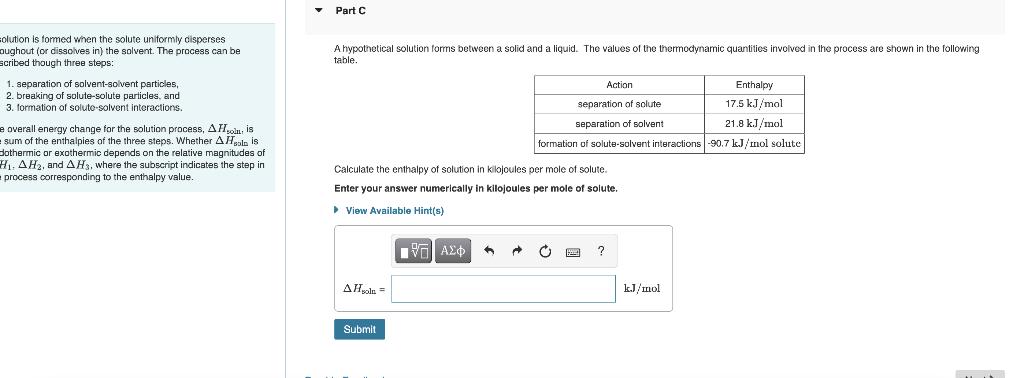
solution is formed when the solute uniformly disperses oughout (or dissolves in) the solvent. The process can be scribed though three steps: 1. separation of solvent-solvent particles, 2. breaking of solute-solute particles, and 3. formation of soluto-solvent interactions. e overall energy change for the solution process, A His sum of the enthalpies of the three steps. Whether A Hol is dothermic or exothermic depends on the relative magnitudes of H₁. AH2, and AH3, where the subscript indicates the step in process corresponding to the enthalpy value. Part C A hypothetical solution forms between a solid and a liquid. The values of the thermodynamic quantities involved in the process are shown in the following table. Action Enthalpy 17.5 kJ/mol separation. solute separation of solvent 21.8 kJ/mol formation of solute-solvent interactions -90.7 kJ/mol solute Calculate the enthalpy of solution in kilojoules per mole of solute. Enter your answer numerically in kilojoules per mole View Available Hint(s) solute. ΤΕΙ ΑΣΦ A C ? AHsola = Submit kJ/mol solution is formed when the solute uniformly disperses oughout (or dissolves in) the solvent. The process can be scribed though three steps: 1. separation of solvent-solvent particles, 2. breaking of solute-solute particles, and 3. formation of soluto-solvent interactions. e overall energy change for the solution process, A His sum of the enthalpies of the three steps. Whether A Hol is dothermic or exothermic depends on the relative magnitudes of H₁. AH2, and AH3, where the subscript indicates the step in process corresponding to the enthalpy value. Part C A hypothetical solution forms between a solid and a liquid. The values of the thermodynamic quantities involved in the process are shown in the following table. Action Enthalpy 17.5 kJ/mol separation. solute separation of solvent 21.8 kJ/mol formation of solute-solvent interactions -90.7 kJ/mol solute Calculate the enthalpy of solution in kilojoules per mole of solute. Enter your answer numerically in kilojoules per mole View Available Hint(s) solute. ΤΕΙ ΑΣΦ A C ? AHsola = Submit kJ/mol
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A secondary rainbow is formed when the incident light undergoes two internal reflections in a spherical drop of water as shown in Fig. 33.20e. (a) In terms of the incident angle Aa A and the...
-
1. A common mirage is formed when the air gets gradually cooler as the height above the ground increases. What might happen if the air grows gradually warmer as the height increases? This often...
-
The standard free energy change for the dissolution of a precipitate with a formula of XCO, is 236 k/mol. Calculate the equilibrium concentration of x" at 25 C. 0.953 M 1,83K10 M 2.07x1021 M...
-
O 4 percent C A TV show on tourism was found to be watched by 9,000 homes. Around 27,000 households that receive the station's broadcasts have TV sets. What is the program rating of the show? 000...
-
Assertions are expressed or implied representations by management that are reflected in the financial statement components. The auditor performs audit procedures to gather evidence to test those...
-
After obtaining an understanding of the client's internal controls, the auditor may choose to test some of the controls. If the results of those tests are satisfactory, the auditor may then rely on...
-
Fill in the Blank. Even a small excitation force can cause an undesirably large response near ____________ .
-
Vice President for Sales and Marketing Sam Totter is trying to plan for the coming year in terms of production needs to meet the sales demand. He is also trying to determine ways in which the...
-
Brian is the IT professional at an electronics company. On a Labor Day weekend where they were offering special sales to customers in selected states, there were a number of complaints that the site...
-
Reported in the table below are estimation results for three residential housing price models. The dependent variable is defined as home sales price measured in thousands of dollars. The models...
-
Consider a Cournot model with 2 firms, where market demand is given by 50-Q if Q50 otherwise 0 P(Q)= = Assume that the firms have different technologies. Firm 2 operates with a modern technology that...
-
Identify the instances of sexual harassment under Title VII of the Civil Rights Act of 1964. Multiple select question. Consensual relationships at the workplace Unwelcome sexual advances...
-
What percentage of adults is considered to be opinion leaders?
-
For the year ended, December 31, 2020, the following information is available for the three product lines that Kincaid, Inc. carries in its inventory: Product Cost Market R89 $108,500 $106,900 N83...
-
Errors in Inventory Count Bow Corp. accidentally overstated its previous year ending inventory by $950. Assume that ending current year inventory is accurately counted. The error in the previous year...
-
Manuel has determined that a fair rate for piecework in his garment factory is $0.70/garment. Determine the gross pay for an employee who produces the following items in one week: Day of Week...
-
3. A firm will pay a dividend of $5 per share next year. Every year after that the dividend is expected to grow at a rate of 2% a year. The beta of the firm's stock is 1.8. R=12% = 5%. What should...
-
The test statistic in the NeymanPearson Lemma and the likelihood ratio test statistic K are intimately related. Consider testing H 0 : = 0 versus H a : = a , and let * denote the test statistic...
-
If untreated, fevers of 104F or higher may lead to brain damage. Why?
-
State whether the sign of the entropy change expected for each of the following processes will be positive or negative, and explain your predictions. (a) PCl3(l) + Cl2(g) PCl5(s) (b) 2HgO(s) 2Hg(l)...
-
Chemical tests of four metals A, B, C, and D show the following results. (a) Only B and C react with 0.5 M HCl to give H2 gas.
-
True or False. The system stiffness matrix is always singular unless the boundary conditions are incorporated.
-
Derive the stiffness and mass matrices of the planar frame element (general beam element) shown in Fig. 12.16 in the global \(X Y\)-coordinate system. Y U U3 U Joint 1 Joint 2 U6 Us UA FIGURE 12.16 A...
-
Fill in the Blank. The displacement within a finite element is expressed in terms of _____________ functions.

Study smarter with the SolutionInn App



