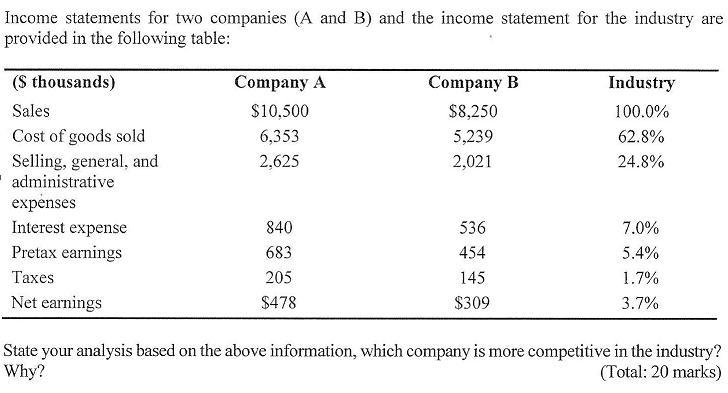
Income statements for two companies (A and B) and the income statement for the industry are...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Income statements for two companies (A and B) and the income statement for the industry are provided in the following table: (S thousands) Sales Cost of goods sold Selling, general, and administrative expenses Interest expense Pretax earnings Taxes Net earnings Company A $10.500 6,353 2,625 840 683 205 $478 Company B $8,250 5,239 2,021 536 454 145 $309 Industry 100.0% 62.8% 24.8% 7.0% 5.4% 1.7% 3.7% State your analysis based on the above information, which company is more competitive in the industry? Why? (Total: 20 marks) Income statements for two companies (A and B) and the income statement for the industry are provided in the following table: (S thousands) Sales Cost of goods sold Selling, general, and administrative expenses Interest expense Pretax earnings Taxes Net earnings Company A $10.500 6,353 2,625 840 683 205 $478 Company B $8,250 5,239 2,021 536 454 145 $309 Industry 100.0% 62.8% 24.8% 7.0% 5.4% 1.7% 3.7% State your analysis based on the above information, which company is more competitive in the industry? Why? (Total: 20 marks)
Expert Answer:
Answer rating: 100% (QA)
We can examine several financial measures and compare them to industry averages to ascertain which c... View the full answer

Related Book For 

Fundamentals of Analytical Chemistry
ISBN: 978-0495558286
9th edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Posted Date:
Students also viewed these chemistry questions
-
A solution was prepared by dissolving 5.76 g of KCl MgCl2 6H2O (277.85 g/mol) in sufficient water to give 2.000 L. Calculate (a) The molar analytical concentration of KCl MgCl2 in this solution....
-
Solution A was prepared by dissolving potassium acetate in methanol. Solution B was prepared by adding potassium methoxide to acetic acid. Reaction of methyl iodide either with solution A or with...
-
A solution was prepared by dissolving 0.834 g of sulfur, S8, in 100.0 g of acetic acid, HC2H3O2. Calculate the freezing point and boiling point of the solution.
-
Thor Bhd. (Thor) is a listed company in Malaysia, specializes in selling batteries. At 31 December 2021, Thor holds four distinct types of batteries in its warehouse. The accountant of Thor provided...
-
What factor or factors make the valuation of common stocks more complicated than the valuation of bonds and preferred stocks?
-
Discuss the challenges that 3PLs and their customers face as they seek to improve performance and reduce costs.
-
Jim Johnson is planning to buy a new car when he graduates from college in three years. He would like to invest a single amount now, in order to have the $24,000 he estimates the car will cost....
-
Sanderson Manufacturing produces ornate, decorative wood frame doors and windows. Each item produced goes through 3 manufacturing processes: cutting, sanding, and finishing. Each door produced...
-
Finance Question. (a) Find the price of a European call option when: 5(0) = 80 K = 80 In the CRR model, the share price in 1 year is either 100 or 60. The risk-free rate is 0 The time horizon is 1...
-
Below is review of the books and records at December 31 for East West College. The review has identified the matters that may need addressing. Record any adjustments required to complete the year end...
-
Cost of supplies are deductible if paid by an ___________ or that were paid for an _____________ and included in their income. Question 23 options: Employee ; Employee Employer ; Employee Employer ;...
-
Requisition Process : A large company is having trouble processing requisition forms for supplies and materials. Just getting through the initial approval process seems to take forever. Then, the...
-
You face an uncertain selling period for your homemade ples. On average, you think you can sell 45 ples. Your costs per pie are $2.50 and each pie sells for $4.50. Any unsold leftovers have no retail...
-
Johnson Company has a target capital structure of 60% equity and 40% debt.Johnson Company currently has 6.5% coupon bonds (with annual coupon payments) outstanding with a par value of $1,000 and 20...
-
Rocker U.S. manufactures low power, low water consuming commercial dishwashers in Malaysia, through their wholly owned subsidiary (MalRoc). MalRocs production cost is $1,650 per unit. MalRoc uses...
-
A collective bargaining session is about to begin. You will represent the Getwell Hospital . (Decide which management position you have) . You will be negotiating with the Hospital Employees Union,...
-
If a firm has a cost of equity of 15 percent, and the firm is 100 percent equity financed. The firm is contemplating a $150 million expansion of its existing operations, funded by selling new stock....
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
Supply the missing data in the table below. Molar Analytical Concentration, Cr Lactic Iodic Butanoic Hypochlorous 0.280 7.00 Nitrous Hydrogen .120 _ 0.200 , 0.640 0.765 5.00 0.644_ __ _0.105 0.413...
-
Calculate the wavelength of the sodium line at 589 nm in an aqueous solution with a refractive index of 1.35.
-
Calculate the pH of a solution that is (a) 0.0100 M in HClO4 and 0.0300 M in monochloroacetic acid. (b) 0.0100 M in HCl and 0.0150 M in H2SO4. (c) 0.0100 M in NaOH and 0.0300 M in Na2S. (d) 0.0100 M...
-
For the following six items, indicate which financial statement category would be affected: (1) net income or (2) other comprehensive income. a. Realized gain on sale of AFS debt investment. b....
-
Refer to Figure 3.4. Suppose Toni's utility function is: Calculate Toni's utility at point Z on Figure 3.4 and compare it with her utility at point M. Which act does Toni prefer? Explain. U;(a)=-160,...
-
What is the beta of: a. The market portfolio b. The risk-free asset c. Portfoho A + B in Example 3.3 and Section 3.7 EXAMPLE 3.3 THE PRINCIPLE OF PORTFOLIO DIVERSIFICATION (PART 2) It turns out that...

Study smarter with the SolutionInn App


