Solve for the volume of 2.5 gallons of juice in units of m. 1 gallon =...
Fantastic news! We've Found the answer you've been seeking!
Question:
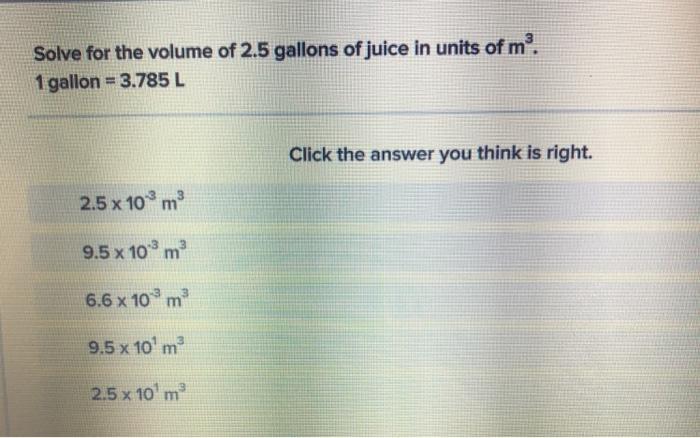
Transcribed Image Text:
Solve for the volume of 2.5 gallons of juice in units of m³. 1 gallon = 3.785 L 3 2.5 x 10 m³ 9.5 x 10³ m³ 6.6 x 10³ m³ 3 9.5 x 10¹ m² 3 2.5 x 10¹ m² Click the answer you think is right. Solve for the volume of 2.5 gallons of juice in units of m³. 1 gallon = 3.785 L 3 2.5 x 10 m³ 9.5 x 10³ m³ 6.6 x 10³ m³ 3 9.5 x 10¹ m² 3 2.5 x 10¹ m² Click the answer you think is right.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
For a simple cubic array, solve for the volume of an interior sphere (cubic hole) in terms of the radius of a sphere in the array.
-
The correct equation for the volume of a sphere is V = 4r3/3, where r is the radius of the sphere. Is the equation in Exercise 12 correct? If not, what should it be when expressed in terms of d?
-
The formula for the volume of a parallelepiped derived in Example 4 should not depend on the choice of which one of the tree vectors we call a, which one we call b, and which one we call c. Use this...
-
In a LVDT, the two secondary voltages a. Are independent of the core position b. Are always in phase quadrature Oc. Vary equally depending on the core position O d. Vary unequally depending on the...
-
Ashton wants to store the following data about S&Ss purchases of inventory: Item number Date of purchase Vendor number Vendor address Vendor name Purchase price Quantity purchased Employee number...
-
In Figure 19.6, the optimum \(\left(E_{0}, C_{0} ight)\) is shown as the point at which the marginal rate of transformation of \(E\) for \(C\) equals the consumer's marginal rate of substitution of...
-
Washington Tennis & Education Foundation, Inc. (WTEF) is a nonprofit organization operating in the District of Columbia that provides athletic and academic programs for children from low-income...
-
Revenue Recognition on Marina Sales with Discounts Taylor Marina has 300 available slips that rent for $800 per season. Payments must be made in full at the start of the boating season, April 1,...
-
When a company lends money to employees at a rate of 4%, the company will record ______. Multiple choice question. a liability called Accounts Payable a liability called Notes Payable an asset called...
-
You have just been hired as a brand manager at Kelsey-White, an American multinational consumer goods company. Recently the firm invested in the development of K-W Vision, a series of systems and...
-
The vending machine subsidies raised a variety of accounting issues for KO and its bottlers: a. How should KO account for this payment? b. How should the anchor bottlers account for the payment? c....
-
Explain the difference between: a cash sale and a credit sale a cash purchase and a credit purchase.
-
You are required to complete the accounting equation formats shown in the Workbook where a business commenced with: a assets of cash at bank $12 000, motor vehicle $25 000 and owners equity $37 000 b...
-
The Accounting Professional and Ethical Standards Board states that there are five areas in which accountants must display a certain standard of professional conduct or ethics. What are they? Explain...
-
Write the missing word(s) in the spaces provided, using the following words: duplicate copy, cheque, control, duplicate receipt, memo, original, receipt, receives, sends, source. a An originating or...
-
Identify, from the suppliers point of view, a source document and the information that should be used included on the document for each of the following transactions: the supplier receives cash from...
-
The expected return on the market portfolio is 12%. The current risk-free rate is 6%. What is the expected return on a stock with a beta of 0.66?
-
U.S. households have become smaller over the years. The following table from the 2010 GSS contains information on the number of people currently aged 18 years or older living in a respondent's...
-
Consider a galvanic cell at standard conditions based on the following half reactions: Au 3+ + 3e - Au o = 1.50 V Fe 3+ + e - Fe 2+ o = 0.77 V When enough NaCl(s) is added to the compartment...
-
It takes 15 kWh (kilowatt hours) of electrical energy to produce 1.0 kg of aluminum metal from aluminum oxide by the HallHeroult process. Compare this value with the amount of energy necessary to...
-
Oxidation of an aldehyde yields a carboxylic acid: Draw the structures for the products of the following oxidation reactions. a. b. c. [ox] propanal 2,3-dimethylpentanal ox] 3-ethylbenzaldehyde>
-
Visit the Suzy Lamplugh Trust website at http://www.suzylamplugh.org and the Social Research Association at http://the-sra.org.uk/sra_resources/safety-code/ . Browse the guidance leaflets/web pages...
-
Andrew, a final year part-time MBA student employed by a large Maltese market research company, read in a local newspaper that Malta (a small island state situated in the centre of the Mediterranean...
-
Maria had a problem. The research proposal for her masters research project was due. But she had no idea what to research. Sustainable tourism had been her favourite module on the programme and...

Study smarter with the SolutionInn App



