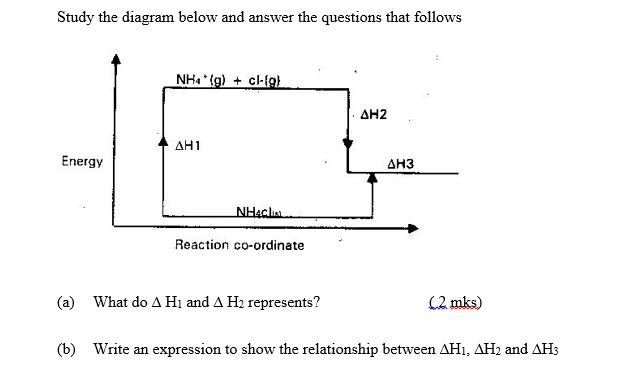
Study the diagram below and answer the questions that follows Energy NH4(g) + cl-(g) AH1 NH4Cl...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Study the diagram below and answer the questions that follows Energy NH4(g) + cl-(g) AH1 NH4Cl Reaction co-ordinate (a) What do A H₁ and A H₂ represents? ΔΗ2 ΔΗ3 (2. mks) (b) Write an expression to show the relationship between AH1, AH2 and AH3 Study the diagram below and answer the questions that follows Energy NH4(g) + cl-(g) AH1 NH4Cl Reaction co-ordinate (a) What do A H₁ and A H₂ represents? ΔΗ2 ΔΗ3 (2. mks) (b) Write an expression to show the relationship between AH1, AH2 and AH3
Expert Answer:
Answer rating: 100% (QA)
a Here H1 and H2 represent the enthalpies of reaction 1 and reaction 2 respectively Enthalpy is r... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemistry questions
-
Study the article below and answer the questions that follow: Drug firm Adcock stays in SA hands Johannesburg -Chile's CFR Pharmaceuticals said on Friday it would drop its $1.2bn bid for drugmaker...
-
Study the article below and answer the questions that follow. Panasonic Scouts U.S. Sites for New Tesla Battery Factory Company eyes locations in Oklahoma, Kansas; talks ongoing Plant to turn out...
-
Study the information given below and answer the questions that follow. Element Atomic radius(nm) Ionic radius nm Formula of oxide Melting point( 0 c) A 0.364 0.421 A 2 O -119 D 0.830 0.711 D O 2 837...
-
A line charge density pL of length L lies parallel to an infinite sheet of surface charge density ps. How much work is required to rotate the line charge so that it is vertical?
-
Describe two disclosures required by the SEC with respect to executive compensation.
-
Discuss in depth the demographer of Germany. Outline the discuss by take in to account the following factors Population, financial status, top 3 diseases and overall healthcare, type of healthcare...
-
In 1982 the Orange County Apartment News carried an advertisement for the eviction services provided by LPS. The ad stated Evictions as low as $65 and showed the picture of a purposeful and...
-
Roxi, Inc., is using a costs-of-quality approach to evaluate design engineering efforts for a new skateboard. Roxi's senior managers expect the engineering work to reduce appraisal, internal failure,...
-
(07.04 MC) When comparing the f(x)=x^(2)+2x and g(x)=log(2x+1), on which interval are both functions negative?
-
Use the following payoff table for Hardaway Corporation and Paxton Industries. These two firms must make simultaneous pricing decisions. They can choose low, medium, or high prices. The payoffs given...
-
Helpful Hardware reports budgeted merchandise purchases below. For those purchases, 40% of a month's purchases is paid in the month of purchase, and 60% is paid in the first month after the purchase....
-
The following data relate to Heavenly Manufacturing Ltd: During July, 133500 units were completed and transferred out. Work in process, 1 July Direct material Conversion Costs incurred during July...
-
what extent do cultural studies methodologies, informed by critical theory and interdisciplinary scholarship, facilitate a nuanced analysis of the production, circulation, and reception of cultural...
-
Briefly describe what cognitive psychology topic is being represented or discussed and how the cognitive psychology topic is being portrayed. Is this topic being represented accurately? Why or why...
-
There is another great example in our text to illustrate shaping. It has to do with a boy, Andrew, who did not speak. A reinforcer, gum, was used to help Andrew begin to speak. If an individual...
-
The SLM Co. is planning on investing in a new project. This will involve the purchase of some new machinery costing $350,000. The project is expected to generate cash inflows of $125,450 each year...
-
You invest $100 in a risky asset with an expected rate of return of 0.14 and a standard deviation of 0.17 and a T-bill with a rate of return of 0.04. To form a portfolio with an expected return of...
-
The vapor pressure of the liquid NH, is measured at different temperatures. The following vapor pressure data are obtained. Temperature, K P, mmHg 217.1 223.4 234.7 588.1 Calculate the enthalpy of...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family. Ivan and Irene have the following investment income, in addition to that reported in Chapter 1: Dividends...
-
Tom has a successful business with $100,000 of income in 2012. He purchases one new asset in 2012, a new machine which is 7-year MACRS property and costs $25,000. If you are Tom's tax advisor, how...
-
Leslie is a single taxpayer who is under age 65 and in good health. For 2012, she has a salary of $23,000 and itemized deductions of $1,000. Leslie is entitled to one exemption on her tax return. a....
-
What kinds of foreign exchange risk would a U.S. company face that is exporting goods to Mexico? What kinds of things would you monitor to determine what will happen to the value of the Mexican peso?
-
Extract the annual report of a Fortune Global 500 company from a country different from your home country; use the site www.glgc.com/g600.html, or...
-
Plan a study term abroad. What would you want to learn and why? How would this change if you were a business?

Study smarter with the SolutionInn App


