Suppose that the market return is 13.3% per year and the risk free rate averaged 4%....
Fantastic news! We've Found the answer you've been seeking!
Question:
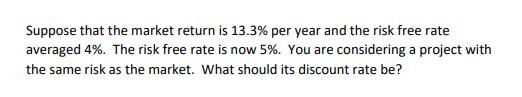
Transcribed Image Text:
Suppose that the market return is 13.3% per year and the risk free rate averaged 4%. The risk free rate is now 5%. You are considering a project with the same risk as the market. What should its discount rate be? Suppose that the market return is 13.3% per year and the risk free rate averaged 4%. The risk free rate is now 5%. You are considering a project with the same risk as the market. What should its discount rate be?
Expert Answer:
Answer rating: 100% (QA)
To determine the discount rate for a project with the ... View the full answer

Related Book For 

Posted Date:
Students also viewed these finance questions
-
What is the NPV for the following project cash flows at a discount rate of 15%? C0=-1,000, C1=700, C2=700 SOB Floor Company has a risk premium of 21% and a beta of 3. If the risk free rate is 3%,...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
You have been hired as a management consultant by AD Corporation to evaluate whether it has an appropriate amount of debt (the company is worried about a lever aged buyout). You have collected the...
-
DFI of real sequences with odd harmonics only Let x(n) be an N -point real sequence with N-point DFT X(k) (N even). In addition, x(n) satisfied the following symmetry property: x(n + N/2) = -x(n) n =...
-
In Prob. 17.89, determine (a) The required distance h if the impulsive reaction at point A is to be zero, (b) The corresponding velocity of the mass center G of the plate immediately after the bullet...
-
The transistor in Figure 6.3 has parameters \(\beta=120\) and \(V_{A}=\infty\). The circuit parameters are \(V_{C C}=3.3 \mathrm{~V}, R_{C}=15 \mathrm{k} \Omega\), and \(I_{C Q}=0.12 \mathrm{~mA}\)....
-
Which depreciation method usually produces the most depreciation in the first year? a. Straight-line b. Units-of-production c. Double-declining-balance d. All produce the same amount of depreciation...
-
Given the following balance sheet, income statement, historical ratios and industry averages, calculate the Pulp, Paper, and Paperboard, Inc. financial ratios for the most recent year. Analyze its...
-
A client maintains a large data center where accesd is limited to authorized employees. How may an auditor best determine the effectiveness of this control activity?
-
Wolsey Industries Inc. expects to maintain the same inventories at the end of 20Y3 as at the beginning of the year. The total of all production costs for the year is therefore assumed to be equal to...
-
A right regular pyramid with height h and a base consisting of a regular n- sided polygon of side length s has a volume V = Ah and total surface area S = A + inst where A is the base area and / the...
-
How would you define the concept of welfare? If well-being is about happiness and satisfaction with life, how would you, as an economist, go about trying to define and quantify happiness and...
-
The following results refer to a test on a petrol engine, indicated power \(=42 \mathrm{~kW}\) Brake power \(\quad=35 \mathrm{~kW}\) Engine speed \(\quad=1800 \mathrm{rpm}\) Fuel consumed per brake...
-
A single cylinder, four stroke C.I. engine has the following data : bore \(=10 \mathrm{~cm} ;\) stroke \(=11 \mathrm{~cm}\), brake torque \(=28 \mathrm{Nm}\), speed \(=1600 \mathrm{rpm} ;\) i.m.e.p....
-
Summarize PathGoal Leadership Theory. What theories of motivation can you tie to the assumptions of the model?
-
Does a correlation between hours of work and nonlabor income measure the income effect?
-
The accounts in the ledger of Treadwell contain the following balances on May 31, 2014. Assuming all balances are normal, determine the missing balance and complete the trial balance below. Accounts...
-
Phosgene, COCl2, is a toxic gas used in the manufacture of urethane plastics. The gas dissociates at high temperature. At 400oC, the equilibrium constant Kc is 8.05 104. Find the percentage of...
-
Now assume that the facts in Problem 1 remain unchanged except for the depreciation method, which is switched to an accelerated method with the following depreciation schedule: Year % of Depreciable...
-
Railroad companies in the United States tend to have long-term, fixed rate, dollar denominated debt. Explain why.
-
Union Pacific Railroad reported net income of $770 million after interest expenses of $320 million in a recent financial year. (The corporate tax rate was 36%.) It reported depreciation of $960...
-
A diesel engine of mass \(300 \mathrm{~kg}\) and operating speed \(1800 \mathrm{rpm}\) is found to have a rotating unbalance of \(1 \mathrm{~kg}-\mathrm{m}\). It is to be installed on the floor of an...
-
A sensitive electronic system, of mass \(30 \mathrm{~kg}\), is supported by a spring-damper system on the floor of a building that is subject to a harmonic motion in the frequency range \(10...
-
A machine of mass \(1150 \mathrm{~kg}\) is mounted on springs. A piston of mass \(m=25 \mathrm{~kg}\) moves up and down in the machine at a speed of \(600 \mathrm{rpm}\) with a stroke of \(350...

Study smarter with the SolutionInn App


