Symbol Name 17C 13C 22Ne 345 Ca Kr 94Zr carbon-12 carbon-13 neon-22 neon-21 sulfur-34 chlorine-37 krypton-82...
Fantastic news! We've Found the answer you've been seeking!
Question:
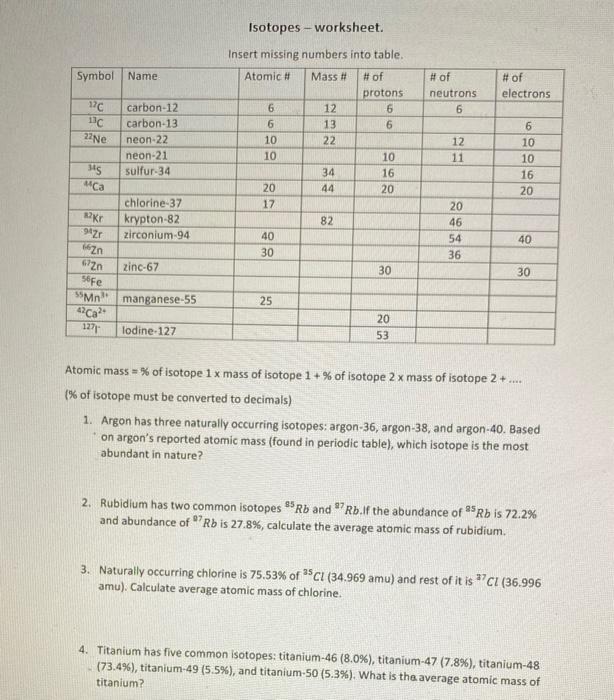
Transcribed Image Text:
Symbol Name 17C 13C 22Ne 345 Ca Kr 94Zr carbon-12 carbon-13 neon-22 neon-21 sulfur-34 chlorine-37 krypton-82 zirconium-94 Zn 672n 56Fe 55Mn 42Ca2+ 1271 lodine-127 zinc-67 manganese-55 Isotopes - worksheet. Insert missing numbers into table. Atomic # Mass # # of protons 6 6 6 10 10 20 17 40 30 25 232 12 13 34 44 82 6 10 16 20 30 20 53 # of neutrons 6 12 11 20 46 54 36 # of electrons 6 10 10 16 20 40 30 Atomic mass=% of isotope 1 x mass of isotope 1+% of isotope 2 x mass of isotope 2+.... (% of isotope must be converted to decimals) 1. Argon has three naturally occurring isotopes: argon-36, argon-38, and argon-40. Based on argon's reported atomic mass (found in periodic table), which isotope is the most abundant in nature? 2. Rubidium has two common isotopes 85Rb and 7Rb.If the abundance of 25 Rb is 72.2% and abundance of 7 Rb is 27.8%, calculate the average atomic mass of rubidium. 3. Naturally occurring chlorine is 75.53% of 35Cl (34.969 amu) and rest of it is 7 Cl (36.996 amu). Calculate average atomic mass of chlorine. 4. Titanium has five common isotopes: titanium-46 (8.0%), titanium-47 (7.8%), titanium-48 (73.4%), titanium-49 (5.5%), and titanium-50 (5.3%). What is the average atomic mass of titanium? Symbol Name 17C 13C 22Ne 345 Ca Kr 94Zr carbon-12 carbon-13 neon-22 neon-21 sulfur-34 chlorine-37 krypton-82 zirconium-94 Zn 672n 56Fe 55Mn 42Ca2+ 1271 lodine-127 zinc-67 manganese-55 Isotopes - worksheet. Insert missing numbers into table. Atomic # Mass # # of protons 6 6 6 10 10 20 17 40 30 25 232 12 13 34 44 82 6 10 16 20 30 20 53 # of neutrons 6 12 11 20 46 54 36 # of electrons 6 10 10 16 20 40 30 Atomic mass=% of isotope 1 x mass of isotope 1+% of isotope 2 x mass of isotope 2+.... (% of isotope must be converted to decimals) 1. Argon has three naturally occurring isotopes: argon-36, argon-38, and argon-40. Based on argon's reported atomic mass (found in periodic table), which isotope is the most abundant in nature? 2. Rubidium has two common isotopes 85Rb and 7Rb.If the abundance of 25 Rb is 72.2% and abundance of 7 Rb is 27.8%, calculate the average atomic mass of rubidium. 3. Naturally occurring chlorine is 75.53% of 35Cl (34.969 amu) and rest of it is 7 Cl (36.996 amu). Calculate average atomic mass of chlorine. 4. Titanium has five common isotopes: titanium-46 (8.0%), titanium-47 (7.8%), titanium-48 (73.4%), titanium-49 (5.5%), and titanium-50 (5.3%). What is the average atomic mass of titanium?
Expert Answer:
Answer rating: 100% (QA)
Answer Symbol Name 17C 13C 22Ne 21 Ne 345 MCa 37 CI 82Kr 94Zr 66Zn 672n 56FC 55Mn 42Ca2 1271 carbon1... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The element oxygen has three naturally occurring isotopes, with 8, 9, and 10 neutrons in the nucleus, respectively. (a) Write the full chemical symbols for these three isotopes. (b) Describe the...
-
Of the isomeric C5H11+ carbocations, which one is the most stable?
-
Which currency is the most widely preferred currency of exchange in global financial markets? Why?
-
What is path dependence? Explain the concept using your own words and provide an example of path dependence (other than what we have already mentioned in class). List the 4 categories of innovation,...
-
The cash payments and purchases journals for Silver Spring Landscaping Co. are shown below. The accounts payable control account has a June 1, 2008 balance of $2,940, consisting of an amount owed to...
-
Given the regression equation Y = -50 + 12X a. What is the change in Y when X changes by +3? b. What is the change in Y when X changes by -4? c. What is the predicted value of Y when X = 12? d. What...
-
An anonymous e-mail is sent to an internal auditor that there is fraud in the inventory/ warehousing cycle. Suggest some appropriate audit steps.
-
Victoria Lee Company had the following adjusted trial balance. Instructions (a) Prepare closing entries at June 30, 2017. (b) Prepare a post-closing trialbalance. VICTORIA LEE COMPANY Adjusted Trial...
-
Saved Help Save & Exit Submit 10 Wickland Company installs a manufacturing machine in its production facility at the beginning of the year at a cost of $101,000. The machine's useful life is estim...
-
In early 2016, Doc and Lyn McGee formed the McGee Cake Company. The company produced a full line of cakes, and its specialties included chess cake,* lemon pound cake, and double-iced, double...
-
Of the following, who is least likely to commit financial statement fraud? senior management board members mid and lower level employees organized criminals
-
How does the hierarchy of decision making relate to these four categories of opera-tions: production, project, event, and incident management?
-
Which is more important, strategic or tactical decision making?
-
Ethics are principles that define what?
-
Find the percentile that corresponds to an age of 57 years old. Use the data set, which represents the ages of 30 executives. 43 57 65 47 57 41 56 53 61 54 56 50 66 56 50 61 47 40 50 43 54 41 48 45...
-
Does a service operation usually experience higher or lower variability in its inputs and outputs, when compared to goods production?
-
Color constancy allows our brain to correct for what feature of vision? A)The persistence of afterimages depends on the spectrum of the light source. B)The light spectrum reflected by an object...
-
Write each fraction as a percent. 7 50
-
A 2.17-g sample of barium reacted completely with water. What is the equation for the reaction? How many milliliters of dry H 2 evolved at 21C and 748 mmHg?
-
An electron is accelerated through a potential difference of 15.6 kilovolts (giving the electron a kinetic energy of 15.6 keV). What is the associated wavelength of the electron in angstroms?
-
Chloroform, CHCl 3 , is a solvent and has been used as an anesthetic. The heat of vaporization of chloroform at its boiling point (61.2C) is 29.6 kJ/mol. What is the entropy change when 5.10 mol CHCl...
-
Explain why a large, nationally chartered bank may be the safest place to deposit your money.
-
Why do you think a law was passed to standardize the ways in which financial institutions inform consumers of the terms and conditions of savings plans?
-
Matthew Delarosa has just moved to a new town and is about to open a checking account. He pays for all his monthly expensesrent, phone, car payment, credit card bills, dry cleaning, and insuranceby...

Study smarter with the SolutionInn App


