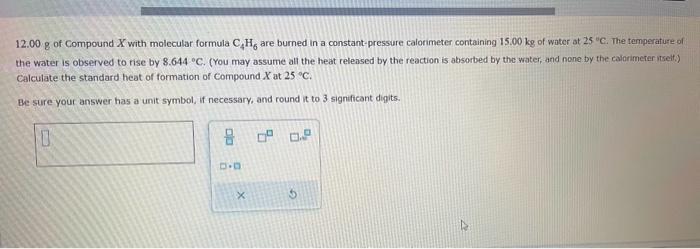
12.00 g of Compound X with molecular formula CH, are burned in a constant-pressure calorimeter containing...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
12.00 g of Compound X with molecular formula C₂H, are burned in a constant-pressure calorimeter containing 15.00 kg of water at 25 °C. The temperature of the water is observed to rise by 8.644 °C. (You may assume all the heat released by the reaction is absorbed by the water, and none by the calorimeter itself.) Calculate the standard heat of formation of Compound X at 25 °C. Be sure your answer has a unit symbol, if necessary, and round it to 3 significant digits. 0 8 D.D X 0.² 0.2 5 12.00 g of Compound X with molecular formula C₂H, are burned in a constant-pressure calorimeter containing 15.00 kg of water at 25 °C. The temperature of the water is observed to rise by 8.644 °C. (You may assume all the heat released by the reaction is absorbed by the water, and none by the calorimeter itself.) Calculate the standard heat of formation of Compound X at 25 °C. Be sure your answer has a unit symbol, if necessary, and round it to 3 significant digits. 0 8 D.D X 0.² 0.2 5
Expert Answer:
Answer rating: 100% (QA)
Here heat released in reaction is absorved by water So Heat gained by water 9 wa... View the full answer

Related Book For 

Accounting Principles
ISBN: 978-1118875056
12th edition
Authors: Jerry Weygandt, Paul Kimmel, Donald Kieso
Posted Date:
Students also viewed these accounting questions
-
Tabares Corporation had these transactions during 2014. (a) Issued $50,000 par value common stock for cash. (b) Purchased a machine for $30,000, giving a long-term note in exchange. (c) Issued...
-
Tabares Corporation had these transactions during 2017. (a) Issued $50,000 par value common stock for cash. (b) Purchased a machine for $30,000, giving a long-term note in exchange. (c) Issued...
-
Tabares Corporation had these transactions during 2019. (a) Issued $50,000 par value common stock for cash. (b) Purchased a machine for $30,000, giving a long-term note in exchange. (c) Issued...
-
You require inventory and accounts receivable collateral for all C&I loans. You have a guideline of an advance rate of 70% for customer receivables of less than 60 days of age. Older receivables get...
-
A homeowner has asked a local real estate agent for advice on the price he should set for his house. The real estate agent notes that the only comparable house in the neighborhood that is currently...
-
If a pattern is used to model an overly broad portion of a system, the generality of resulting pattern is sacrificed. Explain with the help of an example.
-
List the professional requirements for internal environmental auditors and external environmental verifiers and identify the groups which may be equipped to undertake this work.
-
1. What is the nature of the international business environments Harley faces? What types of risks does the firm face? 2. How can Harley benefit from expanding abroad? What types of advantages can...
-
Evaluate how well HRM initiatives contribute to organizational goals and pinpoint any elements that can obstruct their accomplishment.
-
There is an array A made of N integers. Your task is to choose as many integers from A as possible so that, when they are put in ascending order, all of the differences between all pairs of...
-
For the circuit shown in Figure 2, determine its sketch transfer characteristics. Diodes are modelled as a resistance with R50 0 (for simplicity, we ignore the constant voltage of 0.7 V and just...
-
(a) Let f(x) = 5 + 14x. Find f'(x). h '(x) = (b) Let f(x) = e5+14x. Find f'(x). f'(x) =
-
2 . Call Options Payoff and Profit ( 2 0 points ) A European Call option is selling for $ 3 with an exercise price of $ 5 0 , the underlying at expiration is $ 5 3 . Determine the following option...
-
On January 1, Year 1, Grapefruit Corporation purchased a machine for $41,300,000. Grapefruit's management expects to use the machine for 26,000 hours over the next six years. The estimated residual...
-
Determine the moment in N.m about line OB of the force F. F = 595 N. N 325 mm 150 mm Fr P 350 mm y K 400 mm a. MOB = -76.3 N.m b. MOB = -124.9 N.m c. MOB = -44.6 N.m d. MOB = 76.8 N.m 275 mm X
-
2. Nike Inc. currently has an AA- rated (Standard & Poor's) corporate bond outstanding, with a coupon rate of 3.875%, paid semi-annually, due to mature at 11/01/2045 (assume 21 full years), and...
-
Corporate law Topic :shares https://www.slideshare.net/syafawanimahadi/capital-shares-companies-act-2016 Rayyan Global Sdn Bhd's share capital comprises of RM40, 000 ordinary shares issued at RM5.00...
-
Establish identity. cos( + k) = (-1)k cos , k any integer
-
The general ledger of Hensley Company contained the following Accounts Payable control account (in T-account form). Also shown is the related subsidiary ledger. Instructions (a) Indicate the missing...
-
On January 1, 2017, the ledger of Accardo Company contains the following liability accounts. Accounts Payable ....... $52,000 Sales Taxes Payable....... 7,700 Unearned Service Revenue..... 16,000...
-
Ling Corporation uses the equity method to account for its ownership of 35% of the common stock of Gorman Packing. During 2017, Gorman reported a net income of $80,000 and declares and pays cash...
-
What are the various forms of virtual communication used in modern organizations?
-
What are the types of interpersonal communication?
-
How does one choose between communication methods and handle barriers to effective communication?

Study smarter with the SolutionInn App


