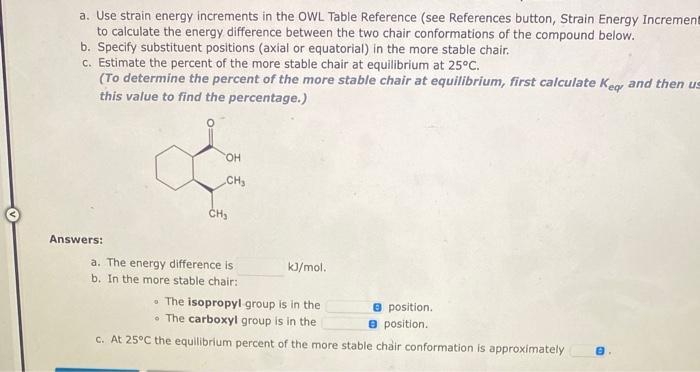
a. Use strain energy increments in the OWL Table Reference (see References button, Strain Energy Increments)...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
a. Use strain energy increments in the OWL Table Reference (see References button, Strain Energy Increments) to calculate the energy difference between the two chair conformations of the compound below. b. Specify substituent positions (axial or equatorial) in the more stable chair. c. Estimate the percent of the more stable chair at equilibrium at 25°C. (To determine the percent of the more stable chair at equilibrium, first calculate Kear and then use this value to find the percentage.) Answers: CH₂ eje H₂C a. The energy difference is b. In the more stable chair: OH kJ/mol. The isopropyl group is in the i • The carboxyl group is in the c. At 25°C the equilibrium percent of the more stable chair conformation is approximately 8 position. 8 position. Use the Références to access important values if needed for this question. a. Use strain energy increments in the OWL Table Reference (see References button, Strain Energy Increments) to calculate the energy difference between the two chair conformations of the compound below. b. Specify substituent positions (axial or equatorial) in the more stable chair. c. Estimate the percent of the more stable chair at equilibrium at 25°C. (To determine the percent of the more stable chair at equilibrium, first calculate Key and then use this value to find the percentage.) Answers: H C=CH₂ CH₂ a. The energy difference is b. In the more stable chair: kJ/mol. • The vinyl(ethenyl) group is in the The methyl group is in the e position. a position. c. At 25°C the equilibrium percent of the more stable chair conformation is approximately 8 a. Use strain energy increments in the OWL Table Reference (see References button, Strain Energy Increment to calculate the energy difference between the two chair conformations of the compound below. b. Specify substituent positions (axial or equatorial) in the more stable chair. c. Estimate the percent of the more stable chair at equilibrium at 25°C. (To determine the percent of the more stable chair at equilibrium, first calculate Key and then us this value to find the percentage.) Answers: OH CH₂ CH₂ a. The energy difference is. b. In the more stable chair: kJ/mol. • The isopropyl group is in the • The carboxyl group is in the c. At 25°C the equilibrium percent of the more stable chair conformation is approximately e position. e position. G a. Use strain energy increments in the OWL Table Reference (see References button, Strain Energy Increments) to calculate the energy difference between the two chair conformations of the compound below. b. Specify substituent positions (axial or equatorial) in the more stable chair. c. Estimate the percent of the more stable chair at equilibrium at 25°C. (To determine the percent of the more stable chair at equilibrium, first calculate Kear and then use this value to find the percentage.) Answers: CH₂ eje H₂C a. The energy difference is b. In the more stable chair: OH kJ/mol. The isopropyl group is in the i • The carboxyl group is in the c. At 25°C the equilibrium percent of the more stable chair conformation is approximately 8 position. 8 position. Use the Références to access important values if needed for this question. a. Use strain energy increments in the OWL Table Reference (see References button, Strain Energy Increments) to calculate the energy difference between the two chair conformations of the compound below. b. Specify substituent positions (axial or equatorial) in the more stable chair. c. Estimate the percent of the more stable chair at equilibrium at 25°C. (To determine the percent of the more stable chair at equilibrium, first calculate Key and then use this value to find the percentage.) Answers: H C=CH₂ CH₂ a. The energy difference is b. In the more stable chair: kJ/mol. • The vinyl(ethenyl) group is in the The methyl group is in the e position. a position. c. At 25°C the equilibrium percent of the more stable chair conformation is approximately 8 a. Use strain energy increments in the OWL Table Reference (see References button, Strain Energy Increment to calculate the energy difference between the two chair conformations of the compound below. b. Specify substituent positions (axial or equatorial) in the more stable chair. c. Estimate the percent of the more stable chair at equilibrium at 25°C. (To determine the percent of the more stable chair at equilibrium, first calculate Key and then us this value to find the percentage.) Answers: OH CH₂ CH₂ a. The energy difference is. b. In the more stable chair: kJ/mol. • The isopropyl group is in the • The carboxyl group is in the c. At 25°C the equilibrium percent of the more stable chair conformation is approximately e position. e position. G
Expert Answer:
Answer rating: 100% (QA)
acHay Energies CH Cving This com be written as two conforr... View the full answer

Related Book For 

Foundations of Finance
ISBN: 9780135160619
10th edition
Authors: Arthur J. Keown, John H. Martin, J. William Petty
Posted Date:
Students also viewed these chemistry questions
-
A small manufacturing firm is considering the purchase of a new boring machine to modernize one of its production lines. Two types of boring machine are available on the market. The lives of machine...
-
A firm is considering the purchase of a new machine to increase the productivity of existing production process. All the alternatives have a life of 10 years and they have negligible market value...
-
A firm is considering the purchase of a new equipment costing $5,052,025 which qualifies for a 26% CCA rate. This equipment has a 4-year life after which it can be sold for $721,980. The firm can...
-
Why are services important? Why is manufacturing important? What are nonmanufactured goods?
-
How do capacity choices vary in the near and long terms?
-
Dr. Zhivgo Diagnostics Corp. income statements for 20X1 are as follows: Sales $2,790,000 Cost of goods sold 1,790,000 Gross profit 1,000,000 Selling and administrative expense 302,000 Operating...
-
A simplified model of a motorcycle traveling over a rough road is shown in Fig. 14.31. It is assumed that the wheel is rigid, the wheel does not leave the road surface, and the cycle moves at a...
-
The following items were taken from the financial statements of D. Gygi Company. (All amounts are in thousands.) Instructions Prepare a classified balance sheet in good form as of December 31,2014....
-
Problem #3 (20 points): A scientist in a laboratory ensemble a group of three charged particles q = 6.0 nC, q2 = 4.0 nC, and q3=-2.0 nC in a formation as shown in the picture to the right. If a = 2.0...
-
Use the 3 step Mathematical Induction process to prove the following statements. You must label and show all three steps. Step 1: Prove n = 1 is true Step 2: Assume true for all n = k Step 3: plug in...
-
RFP to Determine Consumers' Perceptions of Purchasing Multi-Purpose Cleansing and Facial Device The use of skincare devices is on the rise, although only a few Canadians have enjoyed the experience...
-
Part One: Le-Nature's Inc. Case Explain the corporate governance-related responsibilities of the internal roles (accountants) within the case. Explain the corporate governance-related...
-
Modern Electronics Company purchases merchandise inventory from several suppliers. On April 1, 2023, Modern Electronics purchased from Speedy Supplies $120,000 of inventory on account. On May 15,...
-
industry comparison of delta airlines and American Airlines financial statement analysis to determine which company you should invest Provide an analysis of the horizontal, common size, and ratio...
-
based on coco cola income statement https://finance.yahoo.com/quote/KO/financials 1. Profit Margins (Gross Profit Margin, EBIT Margin, Net Profit Margin, EBIDTA Margin, EBITDA Growth, Return on...
-
Justify how a cost-benefit analysis will help to focus the vendor selection process. Discuss the difference between a request for information and a request for a proposal. a.Explain why one might be...
-
Stuart Inc. has sales of $671,000, costs of goods sold $333,000, depreciation expense of $77,000, interest expense of $48,500, a tax rate of 24 percent, and paid out $44,500 in cash dividends. The...
-
The Dow Jones Industrial Average reached a high of $ 7801.63 on December 29, 1997. Recall from Example 18.4 that it reached a high of $ 1003 on November 14, 1972. The Consumer Price Index for...
-
Suppose you purchased 16 shares of Diamond Company stock for $24.22 per share on May 1, 2016. On September 1 of the same year, you sold 12 shares of the stock for $25.68 per share. Calculate the...
-
In 2012 AT&T (T) borrowed $3 billion by issuing bonds in the public bond market. Although this may sound like a lot of money, AT&T owed almost $65 billion in corporate debt at the end of 2011. The...
-
Bill and Kate Theil are not only husband and wife but entrepreneurs who have established three successful businesses. The proposed plan for their latest effort involves a series of international...
-
The phase-plane equation of a single-degree-of-freedom system is given by \[\frac{d y}{d x}=\frac{-c y-\left(x-0.1 x^{3} ight)}{y}\] Investigate the nature of singularity at \((x, y)=(0,0)\) for...
-
Identify the singularity and find the nature of solution near the singularity for van der Pol's equation: \[\ddot{x}-\alpha\left(1-x^{2} ight) \dot{x}+x=0\]
-
Identify the singularity and investigate the nature of solution near the singularity for an undamped system with a hard spring: \[\ddot{x}+\omega_{n}^{2}\left(1+k^{2} x^{2} ight) x=0\]

Study smarter with the SolutionInn App


