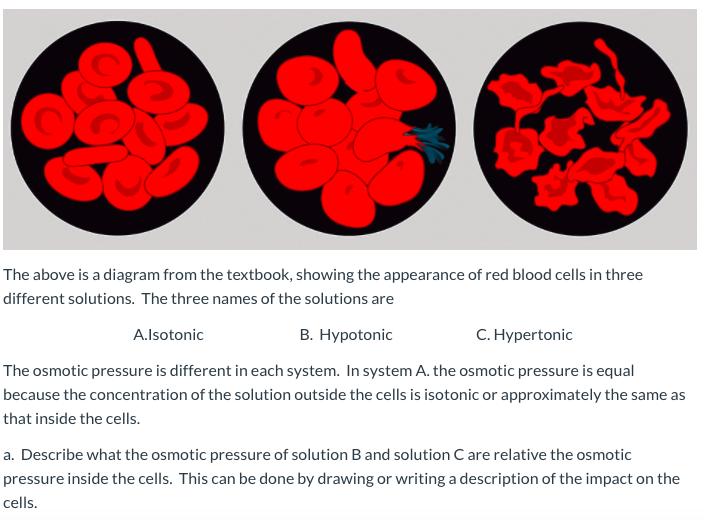
The above is a diagram from the textbook, showing the appearance of red blood cells in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The above is a diagram from the textbook, showing the appearance of red blood cells in three different solutions. The three names of the solutions are В. Нурotonic С. Нурertonic A.lsotonic The osmotic pressure is different in each system. In system A. the osmotic pressure is equal because the concentration of the solution outside the cells is isotonic or approximately the same as that inside the cells. a. Describe what the osmotic pressure of solution B and solution C are relative the osmotic pressure inside the cells. This can be done by drawing or writing a description of the impact on the cells. The above is a diagram from the textbook, showing the appearance of red blood cells in three different solutions. The three names of the solutions are В. Нурotonic С. Нурertonic A.lsotonic The osmotic pressure is different in each system. In system A. the osmotic pressure is equal because the concentration of the solution outside the cells is isotonic or approximately the same as that inside the cells. a. Describe what the osmotic pressure of solution B and solution C are relative the osmotic pressure inside the cells. This can be done by drawing or writing a description of the impact on the cells.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Fig. 1.18 is a picture of red blood cells seen under a scanning electron microscope. Normally, women possess about 4.5 million of these cells in each cubic millimeter of blood. If the blood flow to...
-
Fill in the blank. The production of red blood cells is known as ________________________ which will take ______ days to complete the maturation of the cells before they migrate to the blood vessels.
-
Three prisoners, A, B, and C, are locked in their cells. It is common knowledge that one of them is to be executed the next day and the others are to be pardoned. Only the Governor knows which one...
-
At December 31, 2018, Landy Products has cash of $24,000, receivables of $18,000, and inventory of $80,000. The companys equipment totals $182,000. Landy owes accounts payable of $22,000 and...
-
In this exercise, we examine the rate of sexual violence against females reported by Michael Planty and his colleagues (2013). The table includes the rate of victimization per 1,000 females age 12 or...
-
Explain the meaning of SST, SSR, and SSE, and how they are related to \(R^{2}\).
-
Assuming standard atmospheric conditions, what will happen to your true airspeed when you conduct a constant indicated airspeed climb from sea level to 20,000 feet?
-
Preparing a classified balance sheet Required Use the following information to prepare a classified balance sheet for Little Co. at the end of 2010. Accounts receivable ...... $42,500 Accounts...
-
Discuss the challenges and opportunities of scaling up process intensification concepts from laboratory-scale experiments to industrial-scale applications, addressing issues related to...
-
Taiwan is a major world supplier of semiconductor chips. A recent earthquake severely damaged the production facilities of Taiwanese chip - producing companies, sharply reducing the amount of chips...
-
Two transverse sinusoidal waves combining in a medium are described by the wave functions V = 5.00 sin[(x + 0.300t)] Y25.00 sin[(x -0.300)] where x, y, and y are in centimeters and t is in seconds....
-
You will be required to use on-line research engines to locate a U.S. Supreme Court case and write a brief of the court's opinion using the I.R.A.C. method. You will be expected to provide all three...
-
Information about the assets of TAP Holdings is provided below: TAP purchased land on January 1 , 2 0 1 3 for $ 5 0 0 million. As of January 1 , 2 0 1 8 , the fair value was estimated to be $ 5 5 0...
-
The fiscal year ends December 31 for Lake Hamilton Development. To provide funding for its Moonlight Bay project, LHD issued 7% bonds with a face amount of $600,000 on November 1, 2024. The bonds...
-
Prepare the bank reconciliation statement for the month ending June 30, 2019. Beckett Co. received its bank statement for the month ending June 30, 2019, and reconciled the statement balance to...
-
P1). A centrifugal pump, of 32" diameter, having the characteristics shown in Fig prob. 8 is used to pump water at 60F between two large open tanks through 1500 ft of 16-in.- diameter pipe. The...
-
An individual had the following transactions during 2022 and 2023: item description date proceeds adjusted cost base 1 Sold 1,000 shares of stock A 12 Mar 2022 8,000 6,800 2 Sold 800 shares of stock...
-
The senior management at Davis Watercraft would like to determine if it is possible to improve firm profitability by changing their existing product mix. Currently, the product mix is determined by...
-
A motorist travels 80 km at 100 km/h, and 50 km at 75 km/h. What is the average speed for the trip?
-
Draw schematic diagrams for the electrons in the subshells of (a) Sodium (Na) and (b) Argon (Ar) atoms in the ground state.
-
A diffraction grating is designed to have the second-order maxima at 10o from the central maximum for red light ( = 700 nm). How many lines per centimeter does the grating have?
-
The responses most likely to be associated with use of a force-coercion change strategy are best described as __________. (a) internalized commitment (b) temporary compliance (c) passive cooptation...
-
The assessment center approach to employee selection relies heavily on ____________ to evaluate a candidates job skills. (a) intelligence tests (b) simulations and experiential exercises (c) 360...
-
Which of the following questions can an interviewer legally ask a job candidate during a telephone interview? (a) Are you pregnant or planning to soon start a family? (b) What skills do you have that...

Study smarter with the SolutionInn App


