The acid-catalyzed hydrolysis of sucrose occurs by the following overall reaction whose kinetic data are given below:
Fantastic news! We've Found the answer you've been seeking!
Question:
The acid-catalyzed hydrolysis of sucrose occurs by the following overall reaction whose kinetic data are given below:
C 12 H 2 O 11 (s) +H 2 O (l) ⟶C 6 H 12 O 6 (aq) +C 6 H 12 O 6(aq)
sucrose ⟶ glucose fructose
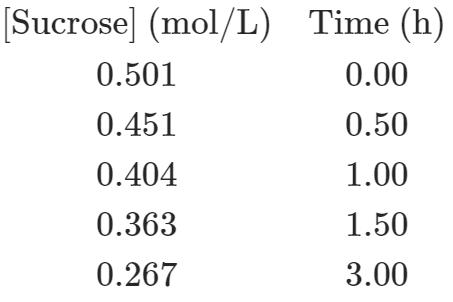
(a) Determine the rate constant and the half-life of the reaction.
(b) How long does it take to hydrolyze 75% of the sucrose?
(c) Other studies have shown that this reaction is actually second-order overall but appears to follow first-order kinetics. (Such a reaction is called a pseudo-first-order reaction.) Suggest a reason for this apparent first-order behavior.
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Posted Date:




