The amount of manganese in steel is determined by changing it to permanganate ion. The steel...
Fantastic news! We've Found the answer you've been seeking!
Question:
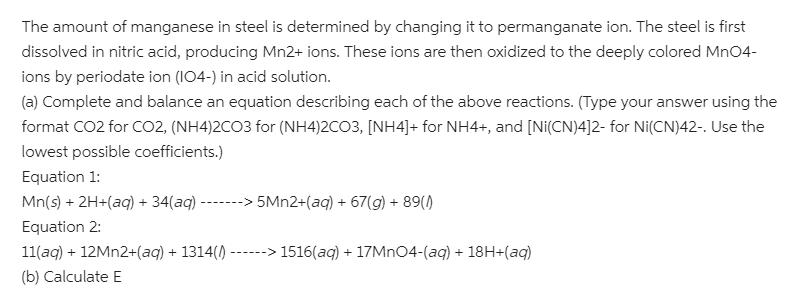
Transcribed Image Text:
The amount of manganese in steel is determined by changing it to permanganate ion. The steel is first dissolved in nitric acid, producing Mn2+ ions. These ions are then oxidized to the deeply colored MnO4- ions by periodate ion (I04-) in acid solution. (a) Complete and balance an equation describing each of the above reactions. (Type your answer using the format CO2 for CO2, (NH4)2CO3 for (NH4)2CO3, [NH4]+ for NH4+, and [Ni(CN)4]2- for Ni(CN)42-. Use the lowest possible coefficients.) Equation 1: Mn(s) + 2H+(aq) + 34(aq) ------> 5MN2+(aq) + 67(g) + 89() Equation 2: 11(aq) + 12MN2+(aq) + 1314() --- ---> 1516(aq) + 17MN04-(aq) + 18H+(aq) (b) Calculate E The amount of manganese in steel is determined by changing it to permanganate ion. The steel is first dissolved in nitric acid, producing Mn2+ ions. These ions are then oxidized to the deeply colored MnO4- ions by periodate ion (I04-) in acid solution. (a) Complete and balance an equation describing each of the above reactions. (Type your answer using the format CO2 for CO2, (NH4)2CO3 for (NH4)2CO3, [NH4]+ for NH4+, and [Ni(CN)4]2- for Ni(CN)42-. Use the lowest possible coefficients.) Equation 1: Mn(s) + 2H+(aq) + 34(aq) ------> 5MN2+(aq) + 67(g) + 89() Equation 2: 11(aq) + 12MN2+(aq) + 1314() --- ---> 1516(aq) + 17MN04-(aq) + 18H+(aq) (b) Calculate E
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Manganese forms three low-spin complex ions with the cyanide ion with the formulas [Mn(CN)6]5-, [Mn(CN)6]4-, and [Mn(CN)6]3-. For each complex ion, determine the oxidation number of Mn and the number...
-
Nitric acid is produced from nitric oxide, NO, which in turn is prepared from ammonia by the Ostwald process: 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(g) What volume of oxygen at 35oC and 2.15 atm is needed...
-
Nitric acid is a strong oxidizing agent. State which of the following species is least likely to be produced when nitric acid reacts with a strong reducing agent such as zinc metal, and explain why:...
-
How did your parents communication with you influence your self-concept?
-
A put option in finance allows you to sell a share of stock at a given price in the future. There are different types of put options. A European put option allows you to sell a share of stock at a...
-
Who might argue for a price ceiling? A price floor?
-
Use the data for Great Gadget, Inc., from 4-32B. Requirements 1. Prepare Great Gadgets multi-step income statement. 2. Calculate the gross profit percentage. 3. The gross profit percentage for 2009...
-
Identify the role and responsibilities that an internal audit staff typically assumes in a company's internal control system.
-
Reorder level (A, B), Two components A and B are used as follows: Normal usage 130 units per week each Minimum usage 145 units per week each Maximum usage 850 units per week each Reorder Quantity A...
-
The Stone River Textile Mill was inspected by OSHA and found to be in violation of a number of safety regulations. The OSHA inspectors ordered the mill to alter some existing machinery to make it...
-
Discuss the major differences between adult and juvenile parole. Do you believe the parole system is a critical part of the corrections process? Explain your position and provide at least two support...
-
Four charges QA = 10.0 C, QB 20.0 C, Qc = 16.0 C, and QD = -16.0 C are placed at the locations shown. Here, d = 2.00 cm, d2 = 5.00 cm, and d3 = 3.00 cm. = Calculate the electric potential Vo at the...
-
Explain the significance of the Sabatier principle in understanding the relationship between catalyst surface binding energies and catalytic activity, particularly in the context of transition metal...
-
Explain two (2 ) challenges when analyzing and interpreting the financial performance of different entities over a specific period of time. (25 marks) Please use credible sources and show all...
-
How do computational methods such as density functional theory (DFT) and molecular dynamics simulations contribute to the rational design and optimization of catalyst materials for specific chemical...
-
A charge + q is located at 6 . 0 0 0 c m , a second charge - 2 q is located at 1 6 . 0 0 c m , and a third charge + q is located at 2 2 . 0 0 c m . The magnitude of a is 2 . 6 5 0 u C . Assume that...
-
Using the Government Finance Officer Association's best principles and elements if the GFOA, does the transportation department budget of Texas follow the "best practices"?
-
If there is an unrealized holding gain on available-for-sale investments, it is reported as?
-
The following phosphorus sulfides are known: P4S3, P4S7, and P4S10. Do these compounds obey the law of multiple proportions?
-
Complete and balance the following equations: (a) K(s) 1 H2O(l) (b) NaH(s) 1 H2O(l) (c) Na(s) 1 O2(g) (d) K(s) 1 O2(g)
-
Consider the following equilibrium systems: Predict the change in the equilibrium constant Kc that would occur in each case if the temperature of the reacting system were raised. (a) A2B (b) A BC (c)...
-
Clausius-Clayperon equation is applicable to (a) Solid-vapour (b) Solid-liquid (c) Liquid-vapour (d) All of these.
-
The reaction \(A(\mathrm{l}) ightarrow R(\mathrm{~g})\) is allowed to reach equilibrium condition in an autoclave. At equilibrium there are two phases - one a pure liquid phase of \(A\) and the other...
-
The equilibrium constant for the reaction \(\mathrm{N}_{2}(\mathrm{~g})+3 \mathrm{H}_{2}(\mathrm{~g}) ightarrow 2 \mathrm{NH}_{3}\) is 0.1084 . Under the same conditions, the equilibrium constant for...

Study smarter with the SolutionInn App


