The binding constants for Mg2+ and Ca2+ are too similar for the species to be determined independently
Fantastic news! We've Found the answer you've been seeking!
Question:
The binding constants for Mg2+ and Ca2+ are too similar for the species to be determined independently by titration without proper treatment. When a dilute NaOH solution is added to the tap water (instead of a buffer), the species can be separated because of the formation of a precipitate. Examine Appendix 2 from Fundamentals of Analytical Chemistry. What is the precipitate that is formed? What species remains in solution and reacts with the EDTA during the titration? What is the name (vocab question) for the role the hydroxide ion (OH-) plays in the process?
appendix 2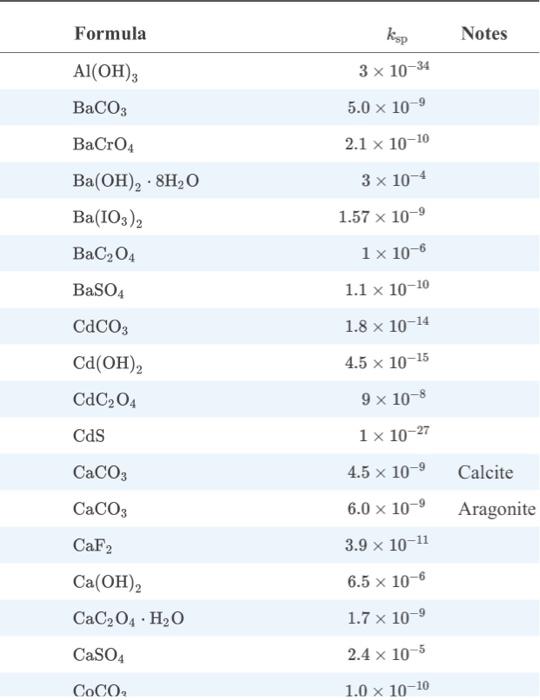
Related Book For 

Fundamentals of Analytical Chemistry
ISBN: 978-0495558286
9th edition
Authors: Douglas A. Skoog,Donald M. West, F. James Holler, Stanley R. Crouch
Posted Date:




