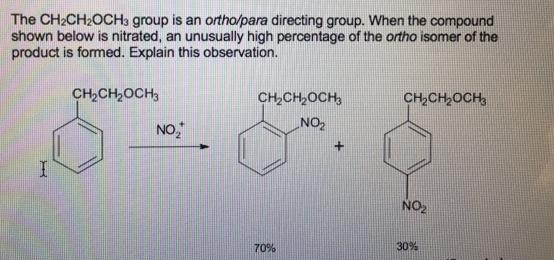
The CH2CH2OCH3 group is an ortho/para directing group. When the compound shown below is nitrated, an...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The CH2CH2OCH3 group is an ortho/para directing group. When the compound shown below is nitrated, an unusually high percentage of the ortho isomer of the product is formed. Explain this observation. CH2CH2OCH3 CH,CH,OCH, CH,CH,OCH NO, ZON NO2 70% 30% The CH2CH2OCH3 group is an ortho/para directing group. When the compound shown below is nitrated, an unusually high percentage of the ortho isomer of the product is formed. Explain this observation. CH2CH2OCH3 CH,CH,OCH, CH,CH,OCH NO, ZON NO2 70% 30%
Expert Answer:
Answer rating: 100% (QA)
1 2 In second question looking at the relative rate for the ortho position in the given ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
When the following compound is heated, a product is formed that shows an infrared absorption band at 1715 cm-1. Draw the structure of the product. CH3 CH
-
Compound B, an isomer of A (Problem 20.47), is also soluble in dilute HCl. The IR spectrum of B shows no bands in the 3300-3500-cm-1 region. The broadband proton-decoupled 13C spectrum of B is given...
-
An ester is a compound formed by a condensation reaction between a carboxylic acid and an alcohol. Read the discussion of esters in Section 24.4 and then give an example of a reaction forming an...
-
An SAT prep course claims to increase student scores by more than 60 points, on average. To test this claim, 9 students who have previously taken the SAT are randomly chosen to take the prep course....
-
An accounting firm uses sampling methods in its client auditing processes. Accounts of a particular type are grouped together in a batch size of 25. The auditor is concerned about erroneous accounts...
-
One kmol of methane (CH4) is burned with an unknown amount of air during a combustion process. If the combustion is complete and there are 2 kmol of free O2 in the products, the airfuel mass ratio is...
-
At what efficiency is a photovoltaic array running if insolation on the collector is \(650 \mathrm{~W} / \mathrm{m}^{2}\), the total collector area is \(10 \mathrm{~m}^{2}\), the voltage across the...
-
Karysa Co. operates in a city in which real estate tax bills for one year are issued in May of the subsequent year. Thus tax bills for 2010 are issued in May 2011 and are payable in July 2011....
-
The Cantina Restaurant in Reno Nevada has just started a new restaurant in Arizona and has asked you to analyze their operation. Their latest month shows the following results: Number of meals...
-
On January 1, 2020, Valley Company entered into a 3-year construction contract that had an estimated gross revenue of P3,000,000. The entity used the percentage of completion in recognizing income...
-
Is it appropriate for the FDA to approve drugs for race-specific indications, as it did with BiDil? What are the potential risks to race-specific drug approvals? What might the FDA (or other...
-
What are the approaches firms can take to create new services?
-
How can a firm design perceived control into a service encounter? Apply it to one face-to-face and one online encounter.
-
What actions could a bank take to encourage more customers to transact via the internet, apps, and ATMs instead of visiting a branch?
-
Give examples of how Internet and telecommunications technologies (e.g., mobile commerce [M-Commerce] and apps) have changed some of the services you use.
-
How is branding used in services marketing? What is the distinction between a corporate brand such as Marriott and the names of its various inn and hotel chains?
-
Variable costs per unit: Manufacturing: Direct materials $ 21 Direct labor $ 13 Variable manufacturing overhead $ 3 Variable selling and administrative $ 2 Fixed costs per year: Fixed manufacturing...
-
Find the velocity, acceleration, and speed of a particle with the given position function. r(t) = (t 2 , sin t - t cos t, cos t + t sin t), t > 0
-
a. How many asymmetric carbons does cholesterol have? b. What is the maximum number of stereoisomers that cholesterol can have? c. How many of these stereoisomers are found in nature? CHj H3C CH3 HC...
-
Draw a reaction coordinate diagram for a reaction in which a. the product is thermodynamically unstable and kinetically unstable. b. the product is thermodynamically unstable and kinetically stable.
-
Explain why thymine cannot be deaminated.
-
The A-36 steel post is subjected to the forces shown. If the strain gages \(a\) and \(b\) at point \(A\) give readings of \(\epsilon_{a}=300\left(10^{-6} ight)\) and \(\epsilon_{b}=175\left(10^{-6}...
-
The state of strain at the point on the bracket has components of \(\epsilon_{x}=-130\left(10^{-6} ight), \quad \epsilon_{y}=280\left(10^{-6} ight)\), \(\gamma_{x y}=75\left(10^{-6} ight)\). Use the...
-
A differential element is subjected to plane strain that has the following components: \(\epsilon_{x}=950\left(10^{-6} ight), \epsilon_{y}=420\left(10^{-6} ight)\), \(\gamma_{x y}=-325\left(10^{-6}...

Study smarter with the SolutionInn App


