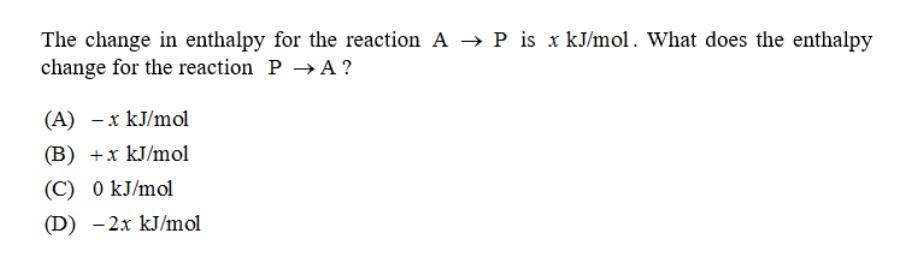The change in enthalpy for the reaction A P is x kJ/mol. What does the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The change in enthalpy for the reaction A → P is x kJ/mol. What does the enthalpy change for the reaction P→A ? (A) -x kJ/mol (B) +x kJ/mol (C) 0 kJ/mol (D) -2x kJ/mol The change in enthalpy for the reaction A → P is x kJ/mol. What does the enthalpy change for the reaction P→A ? (A) -x kJ/mol (B) +x kJ/mol (C) 0 kJ/mol (D) -2x kJ/mol
Expert Answer:
Related Book For 

Fundamentals of Thermodynamics
ISBN: 978-0471152323
6th edition
Authors: Richard E. Sonntag, Claus Borgnakke, Gordon J. Van Wylen
Posted Date:
Students also viewed these chemistry questions
-
For an application the change in enthalpy of carbon dioxide from 30 to 1500C at 100 kPa is needed. Consider the following methods and indicate the most accurate one. a. Constant specific heat, value...
-
For an application the change in enthalpy of carbon dioxide from 30 to 1500C at 100 kPa is needed. Consider the following methods and indicate the most accurate one. a. Constant specific heat, value...
-
For an application the change in enthalpy of carbon dioxide from 30 to 1500C at 100 kPa is needed. Consider the following methods and indicate the most accurate one. a. Constant specific heat, value...
-
Vicky's income for 2020-21 consists of her UK salary of 50,270 and rents from overseas property (net of 30% withholding tax) of 3,500. Vicky is UK resident in 2020-21 and she is not a Scottish...
-
What is the work performed when a gas expands from 0.66 L to 1.33 L against an external pressure of 0.775 atm?
-
Use implicit differentiation to find z/x and z/y. yz = ln(x + z)
-
Organic Co. makes a variety of products from a joint process. The joint cost per batch run is \(\$ 30,000\) and each batch produces the following products: How would you classify each product (joint,...
-
On February 28, 2012, Starfish Corp. issues 10%, five-year bonds payable with a face value of $1,200,000. The bonds pay interest on February 28 and August 31. Starfish Corp. amortizes bonds by the...
-
J&R Construction Company is an international conglomerate with a real estate division that owns the right to erect an office building on a parcel of land in downtown Sacramento over the next year....
-
Rachel Cook is very concerned. Until recently, she has always had the golden touch, having successfully launched two start- up companies that made her a very wealthy woman. However, the timing could...
-
Explain two sources of finance Adriana is unlikely to be able to draw on to fund her aggressive marketing strategies.
-
Refer the following table. Focus Metals Inc. Comparative Balance Sheet Information November 30 (millions of $) 2017 2016 Cash $ 25 $ 95 Accounts receivable (net) 422 268 Inventory 78 71 Plant and...
-
A communications truck with a 46-cm-diameter dish receiver on the roof starts out 12 km from its base station. It drives directly away from the base station at 55 km/h for 1.0 h, keeping the receiver...
-
How does the concept of a "deadlock-free" system align with the practical realities of resource management in operating systems, and what are the potential drawbacks of striving for complete deadlock...
-
A fixed 15.4 cm diameter wire coil is perpendicular to a magnetic field 0.43 T pointing up. In 0.11 s, the field is changed to 0.3 T pointing down. What is the average induced emf in the coil
-
Assume that there are two factors that price assets and there is an asset with no systematic risk that earns a rate of return of 5%. Factor 1 has a risk premium of 5% and factor has a risk premium of...
-
The population mean brain-weight at 90 days of age for a particular strain of inbred mice is 3.36 grams. A group of 25 of these mice are reared in a special environment designed to stimulate diverse...
-
The Pletcher Transportation Company uses a responsibility reporting system to measure the performance of its three investment centers: Planes, Taxis, and Limos. Segment performance is measured using...
-
Calculate the equilibrium constant for the reaction H2 2H at a temperature of 2000 K, using properties from Table A.9. Compare the result with the value listed in Table A.11.
-
A small turbine delivers 150 kW and is supplied with steam at 700C, 2 MPa. The exhaust passes through a heat exchanger where the pressure is 10 kPa and exits as saturated liquid. The turbine is...
-
Fresh water can be produced from saltwater by evaporation and subsequent condensation. An example is shown in Fig. P10.42, where 150-kg/s saltwater, state 1, comes from the condenser in a large power...
-
Suppose you are often dissatisfied with the way things are done in your institution but are afraid to speak up or propose alternative ways of doing things. Does this mean you can never be a leader in...
-
How can one be a leader as a staff nurse?
-
Pittman, Rosenbach, and Potter (1998) described types of followers as: A. Subordinates who do what they are told but who are not actively involved. B. Contributors who are supportive, involved, and...

Study smarter with the SolutionInn App