The electrochemical extraction of aluminum from bauxite ore involves (A) the reaction of AlO3 with coke...
Fantastic news! We've Found the answer you've been seeking!
Question:
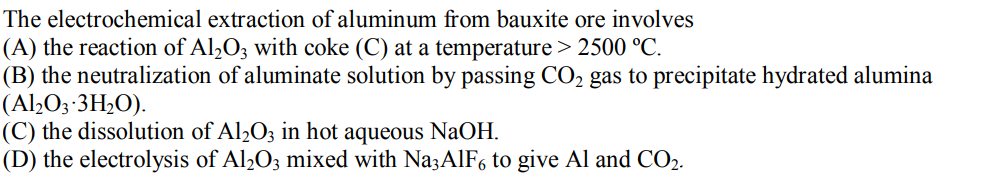
Transcribed Image Text:
The electrochemical extraction of aluminum from bauxite ore involves (A) the reaction of Al₂O3 with coke (C) at a temperature > 2500 °C. (B) the neutralization of aluminate solution by passing CO₂ gas to precipitate hydrated alumina (Al₂O3-3H₂O). (C) the dissolution of Al2O3 in hot aqueous NaOH. (D) the electrolysis of Al2O3 mixed with Na3AlF6 to give Al and CO2. The electrochemical extraction of aluminum from bauxite ore involves (A) the reaction of Al₂O3 with coke (C) at a temperature > 2500 °C. (B) the neutralization of aluminate solution by passing CO₂ gas to precipitate hydrated alumina (Al₂O3-3H₂O). (C) the dissolution of Al2O3 in hot aqueous NaOH. (D) the electrolysis of Al2O3 mixed with Na3AlF6 to give Al and CO2.
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Answer B t... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A mass of 400(lbm) of 35-wl-% aqueous NaOH solution at 130(F) is mixed with 175(lbm) of 10-wt-% solution at 200(F). (a) What is the heat effect if the final temperature is 80(F)? (b) If the mixing is...
-
A carbonated drink is fully charged with CO2 gas at 17C and 600 kPa such that the entire bulk of the drink is in thermodynamic equilibrium with the CO2-water vapor mixture. Now consider a 2-L soda...
-
A carbonated drink is fully charged with CO2 gas at 17C and 600 kPa such that the entire bulk of the drink is in thermodynamic equilibrium with the CO2water vapor mixture. Now consider a 2-L soda...
-
The following is a condensed income statement for Sweets & Treats, a confectionary. Required: Determine (a) The cost of purchases and (b) Costs associated with sales and administration. Sweets &...
-
Solve the following problem using a spreadsheet. Your S corporation needs a new track hoe for its operations and is looking at three alternatives. The first alternative is to lease the track hoe for...
-
The following accounts appear in Theresa Gortons general ledger. (The dates of the transactions have been omitted). Required a) Close down the relevant accounts in the general ledger. b) Show how...
-
Which of the following statements is false? a. Activity-based costing can only be used by manufacturing firms. b. Activity-based costing focuses on allocating indirect costs. c. An activity-based...
-
On January 1, 2016, Brown Co. borrowed cash from First Bank by issuing a $100,000 face value, four-year term note that had an 8 percent annual interest rate. The note is to be repaid by making annual...
-
1. 2. 3. (c) 11 Let f : R R be a continuous function such that f(3x)-f(x) =. If f(8) = 7, then f(14) is equal to: (a) 4 (b) 10 (d) 16 Let O be the origin and A be the point z = 1 + 2i. If B is the...
-
A B E F 1 Better Bottles, Inc., uses a periodic inventory system and has the following information available: 2 Cost per Unit 20 $ 3 Description 4 Beginning Inventory 5 Jan. 15 Purchase 6 Jan. 20...
-
What is one way to enhance the quality of Leader - Member Exchanges ( LMX ) between leaders and their employees and increase team performance?
-
The thermal radiation from the earth's surface peaks at a wavelength of approximately \(10 \mu \mathrm{m}\). What is the energy of a photon at this wavelength? A. \(2.4 \mathrm{eV}\) B. \(1.2...
-
1. What kind of organizational politics are at work here? 2. What influence tactics might the profs have used to get the grad students to fill in for them? 3. Discuss the ethics of the professors...
-
Explain the needs of internal users for management accounting information (section 1.2). 1.2 Meeting the needs of internal users Although the definition of accounting remains appropriate for internal...
-
In Problems 530, a. Classify the sequences as arithmetic, geometric, Fibonacci, or none of these. b. If arithmetic, give d; if geometric, give r; if Fibonacci, give the first two terms; and if none...
-
Bob can throw a \(500 \mathrm{~g}\) rock with a speed of \(30 \mathrm{~m} / \mathrm{s}\). During the INT time the rock is in his hand, his hand moves forward by \(1.0 \mathrm{~m}\). a. How much...
-
Ms Moore owns a 16 unit apartment complex. The market rent on each unit is $500 per month. It is expected that on average, one unit will be vacant for the 3 months a year. The annual operating...
-
What did Lennox gain by integrating their WMS, TMS, and labor management systems?
-
For the distillation operation shown in figure, establish the type of condenser and an operating pressure, calculate the minimum number of equilibrium stages, and estimate the distribution of the...
-
Design a two-column distillation sequence to separate 1,000 kmol/h of 20 mol% aqueous acetic acid into nearly pure acetic acid and nearly pure water. The first column should use heterogeneous...
-
Estimate the liquid diffusivity of acetic acid at 25oC in a dilute solution of:(a) Benzene,(b) Acetone,(c) Ethyl acetate, and(d) Water by an appropriate method. Compare the estimated values with the...
-
Deflation is the rate of decline in the aggregate price level. Why might unexpected deflation be of particular concern to someone managing a bank?
-
Analyze how a government regulates and supervises the financial system to contain risk.
-
One goal of the regulatory reforms that followed the 2007-2009 financial crisis was to address the too-big-to-fail problem associated with large institutions. How did the reforms try to address this...

Study smarter with the SolutionInn App


