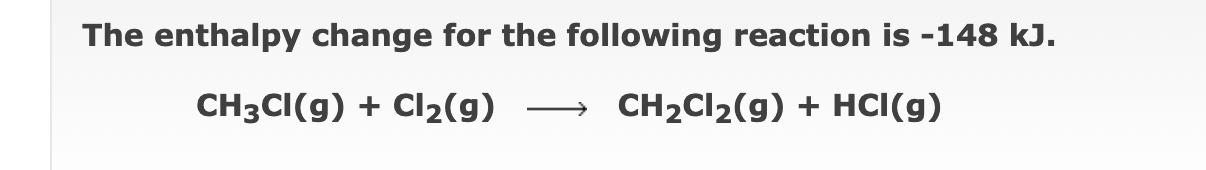
The enthalpy change for the following reaction is -148 kJ. CH3CI(g) + Cl(g) CHCl(g) + HCI(g)...
Fantastic news! We've Found the answer you've been seeking!
Question:





Transcribed Image Text:
The enthalpy change for the following reaction is -148 kJ. CH3CI(g) + Cl₂(g) CH₂Cl₂(g) + HCI(g) b Estimate the C-H bond energy in CH3CI(g), using tabulated bond energies (linked above) for the remaining bonds. C-H bond energy = kJ/mol The enthalpy change for the following reaction is -136 kJ. H₂(g) + O₂(g) H₂O₂(g) b Estimate the H-O bond energy in H₂O₂(g), using tabulated bond energies (linked above) for the remaining bonds. H-O bond energy = kJ/mol The enthalpy change for the following reaction is -566 kJ. 2CO(g) + O₂(g) →→2CO₂(g) b Estimate the O=0 bond energy in O₂(g), using tabulated bond energies (linked above) for the remaining bonds. O=O bond energy = kJ/mol The enthalpy change for the following reaction is -148 kJ. CH3CI(g) + Cl₂(g) CH₂Cl₂(g) + HCI(g) b Estimate the C-H bond energy in CH3CI(g), using tabulated bond energies (linked above) for the remaining bonds. C-H bond energy = kJ/mol The enthalpy change for the following reaction is -136 kJ. H₂(g) + O₂(g) H₂O₂(g) b Estimate the H-O bond energy in H₂O₂(g), using tabulated bond energies (linked above) for the remaining bonds. H-O bond energy = kJ/mol The enthalpy change for the following reaction is -566 kJ. 2CO(g) + O₂(g) →→2CO₂(g) b Estimate the O=0 bond energy in O₂(g), using tabulated bond energies (linked above) for the remaining bonds. O=O bond energy = kJ/mol
Expert Answer:
Answer rating: 100% (QA)
H 14 HCCL CCL HCCL HGH 1 H C1 Bond ralues this problem by taking t... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these physics questions
-
The standard enthalpy change for the following reaction is 436.4 kJ/mol: H2(g) - H(g) + H(g) Calculate the standard enthalpy of formation of atomic hydrogen (H)?
-
Calculate the enthalpy change for the following reaction: 3NO2(g) + H2O(l) 2HNO3(aq) + NO(g) Use standard enthalpies of formation.
-
The enthalpy change for each of the following reactions was calculated using bond energies. The bond energies of XO, YO, and ZO are all equal. XX + O=O XOOX; H = 275 kJ YY + O=O YOOY; H = +275 kJ...
-
Have leadership scholars, over the past 50 years, managed to produce a clear, cookie-cutter profile of the ideal leader through more than 1,000 studies
-
The following section is taken from Zenith Oil Companys balance sheet at December 31, 2013. Interest is payable annually on January 1. The bonds are callable on any annual interest date. Zenith uses...
-
Determine relative weights and perform a weighted adjustment (to the nearest second) for angles A, B, and C of a plane triangle, given the following four observations for each angle: Angle A Angle B...
-
Mikes Powersports uses the (perpetual) LIFO inventory method. Mikes Powersports started August with 10 helmets that cost \($54\) each. On August 19, Mikes Powersports bought 15 helmets at \($56\)...
-
When a dispute arises between an audit client and its auditor regarding the proper accounting treatment for a transaction or other item, the audit client will sometimes retain another accounting firm...
-
Wonder Woman was paying a visit to Batman so she flew her invisible jet over to the Bat Cave and parked it outside. As she approached the Bat Cave the camouflage covering began to open. Wonder Woman...
-
The Precision Machining Company makes hand-held tools on an assembly line that produces one product every minute. On one of the products, the critical quality dimension is the diameter (measured in...
-
Table B.7 of Appendix B gives the following values for steam at 400C and 150 bar: H = 2975 kJ/kg, U = 2744 kJ/kg. V = 0.0157m 3 /kg (a) The value 2975 is not the true specific enthalpy of steam at...
-
Door Dash Economic Growth and Stability Identify the general economic conditions of the country, region, state, or local area where the firm's target customers are located. How are these economic...
-
After reading "A Conservative Profession of Faith", and "A Letter from Mexico" in The Mexico Reader, discuss what do they tell us about foreign relations in the new nation.?
-
Feathered Depot sold 5 7 , 7 5 0 of gift cards during the current year, and recieved cash. Feathered uses the proportional method for accounting for gift card breakage. Based on historical...
-
What are the negotiating steps that are involved in a US Ambassador planning negotiations to oppose a US vote for a new environmental bill in the United Nations?
-
Why was paying back foreign debt so important for the New nation ? And, conversely, why doesn't the US seem to be as preoccupied with paying back foreign debts today?
-
I. Compute Leland's taxable income or loss for Year 1, and determine the amount of income or loss (1) allocable to and (2) deductible by Josie Chamberlain on her Year 1 federal individual income tax...
-
Following is the current balance sheet for a local partnership of doctors: The following questions represent independent situations: a. E is going to invest enough money in this partnership to...
-
A quantity of the blue solid on the left in Figure 13.7 is placed in an oven and heated for a time. It slowly turns from blue to the white of the solid on the right. What has occurred?
-
An ionic compound has a very negative in water. Would you expect it to be very soluble or nearly insoluble in water? Explain in terms of the enthalpy and entropy changes that accompany the process?
-
Calculate the concentration of an aqueous solution of NaOH that has a pH of 11.50.
-
A centrifugal pump impeller having dimensions and angles as shown rotates at \(500 \mathrm{rpm}\). Assuming a radial direction of velocity at the blade entrance, calculate the flow rate, the pressure...
-
Data measured during tests of a centrifugal pump driven at \(3000 \mathrm{rpm}\) are The flow rate is \(65 \mathrm{gpm}\) and the torque applied to the pump shaft is \(4.75 \mathrm{lbf} \cdot...
-
A centrifugal pump impeller having \(r_{1}=50 \mathrm{~mm}\), \(r_{2}=150 \mathrm{~mm}\), and width \(b=3.75 \mathrm{~mm}\) is to pump \(225 \mathrm{~L} / \mathrm{s}\) of water and supply \(12.2...

Study smarter with the SolutionInn App


