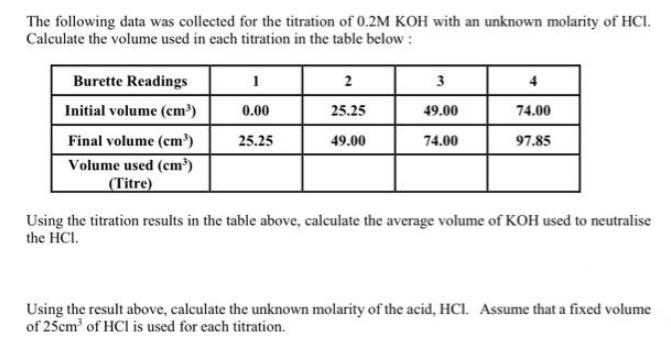
The following data was collected for the titration of 0.2M KOH with an unknown molarity of...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
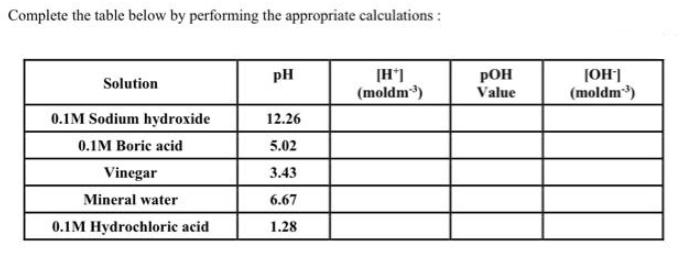
The following data was collected for the titration of 0.2M KOH with an unknown molarity of HCI. Calculate the volume used in each titration in the table below: Burette Readings Initial volume (cm³) Final volume (cm³) Volume used (cm³) (Titre) 1 0.00 25.25 2 25.25 49.00 3 49.00 74.00 4 74.00 97.85 Using the titration results in the table above, calculate the average volume of KOH used to neutralise the HCI. Using the result above, calculate the unknown molarity of the acid, HCI. Assume that a fixed volume of 25cm³ of HCl is used for each titration. Complete the table below by performing the appropriate calculations: Solution 0.1M Sodium hydroxide 0.1M Boric acid Vinegar Mineral water 0.1M Hydrochloric acid PH 12.26 5.02 3.43 6.67 1.28 [H*] (moldm³) POH Value [OH-] (moldm³) Give the following volumes with the appropriate units: (i) 20 21 22 23 24 25 (ii) 28 The following data was collected for the titration of 0.2M KOH with an unknown molarity of HCI. Calculate the volume used in each titration in the table below: Burette Readings Initial volume (cm³) Final volume (cm³) Volume used (cm³) (Titre) 1 0.00 25.25 2 25.25 49.00 3 49.00 74.00 4 74.00 97.85 Using the titration results in the table above, calculate the average volume of KOH used to neutralise the HCI. Using the result above, calculate the unknown molarity of the acid, HCI. Assume that a fixed volume of 25cm³ of HCl is used for each titration. Complete the table below by performing the appropriate calculations: Solution 0.1M Sodium hydroxide 0.1M Boric acid Vinegar Mineral water 0.1M Hydrochloric acid PH 12.26 5.02 3.43 6.67 1.28 [H*] (moldm³) POH Value [OH-] (moldm³) Give the following volumes with the appropriate units: (i) 20 21 22 23 24 25 (ii) 28
Expert Answer:
Answer rating: 100% (QA)
To determine the average volume of KOH used compute the amount utilized in each titration using the ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The highest temperatures ever recorded (in F) in 32 different U.S. states are as follows: 100 109 114 118 100 110 115 119 105 110 116 120 105 112 117 121 106 112 118 122 106 112 118 125 107 114 118...
-
What are the differences between the following two lines in /etc/exports file? /pool 172.16.1.0/24(rw) /pool 172.16.1.0/24 (rw) [4] List exportfs command example usage with the following options. -r:...
-
Pierce Phones is considering the introduction of a new model of headphone whose selling price is $18 per unit and whose variable expense is $15 per unit. The company's monthly fixed expense is...
-
In Exercises 1-4, use the given conditions to find the values of all six trigonometric functions. 1. sec x = - 5 / 2, tan x < 0 2. csc x = - 7 / 6, tan x > 0 3. sin = - 3 / 4, cos > 0 4. cos = 2 /...
-
Use the Binomial Theorem to expand (3x + 1/2 y) 6 .
-
When you remove a dielectric slab from between the plates of a charged isolated capacitor, what happens to the energy stored in the capacitor? Why does this happen to the stored energy?
-
What are the tax consequences to Euclid from the following independent events? a. Euclid bought 500 shares of common stock five years ago for $50,000. This year, Euclid receives 20 shares of common...
-
31 The number of protons, electrons and neutrons in aluminium ion Al+ is Protons A. 27 B. 13 C. ABCD 32 32. D. 13 10 Electron 27 neutrons 14 14 14 10 14 17 14 The formula of the compound formed...
-
7. A retail store sells a jacket for RM290, less 18%. A department store sells the same jacket for RM265, less than 12%. Which store is offering the item for a cheaper price, and for how much? 8....
-
What is the RATIO DECINDENDI for Rowe v. Canning? 1991 ST. J. NO. 0925 IN THE SUPREME COURT OF NEWFOUNDLAND TRIAL DIVISION BETWEEN: BRYAN ROWE (GOODYEAR) AND WAYNE FUREY PLAINTIFFS AND: BOYD CANNING...
-
Jess is a paralegal who received his license four months ago. He works for a senior paralegal named Loralei who is representing the defendant on a complicated collection matter. Jess and Lorelai...
-
Common stock was $525,000 and retained earnings was $1,250,000 as of January 1, 20Y5. Bong there pri additional com During the year, additional common stock of $50,000 was issued for cash, and...
-
In the table below you can see financial information from Haga's shoe department: Sales revenue = ISK 200,000 Variable costs = ISK 140,000 Fixed costs = ISK 100,000 Profit (loss) -40,000 ISK If the...
-
a chart showing the relevant time periods of the offering process including the pre-filing or "quiet period", "the waiting period" and the post effective period. Make a quick and crude list of...
-
At the time of Rana Plaza accident, Bangladesh has employment and labor laws. Were those existing laws insufficient or ineffective? What did you learn from reading the case? Was Bangladesh government...
-
The Strahler Stream Order System ranks streams based on the number of tributaries that have merged. It is a top-down system where rivers of the first order are the headwaters (aka outermost...
-
In an experiment analogous to that in Figure 27-3, the sampling constant is found to be Ks = 20 g. (a) What mass of sample is required for a 2% sampling standard deviation? (b) How many samples of...
-
A solution containing 20.0 mL of 1.00 10 -3 M Co 2+ in the presence of 0.10 M C 2 O 4 2- at pH 9.00 was titrated with 1.00 10 -2 M EDTA. Using formation constants from Appendix I for Co(C 2 O 4 )...
-
Consider the titration of the weak acid HA with NaOH. At what fraction of Ve does pH = pKa - 1? At what fraction of Ve does pH pKa - 1? Use these two points, plus Ve, and 1.2Ve to sketch the...
-
In 1987, the mean salary for major league baseball players was \($412,000.\) In 2011, it was \($3,319,000.\) Compare the increase in mean baseball salaries to the overall rate of inflation as...
-
You want to determine the mean height of all basketball players on the LA Lakers team. Determine whether a census is practical in the situations described. Explain your reasoning.
-
You want to determine the mean age of all statistics instructors at the University of Colorado. representative Samples? In Exercises 1316, identify the sample, population, and sampling method. Then...

Study smarter with the SolutionInn App


