The following information is available from the records of LoopTech Services. On January 1, Velma Morgan,...
Fantastic news! We've Found the answer you've been seeking!
Question:
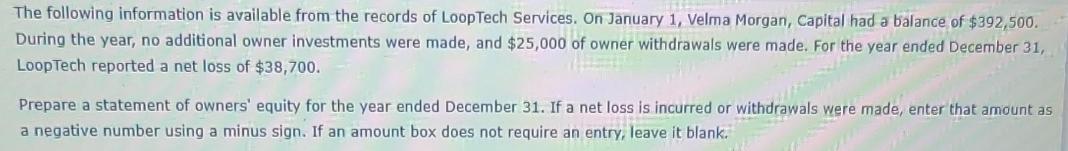
Transcribed Image Text:
The following information is available from the records of LoopTech Services. On January 1, Velma Morgan, Capital had a balance of $392,500. During the year, no additional owner investments were made, and $25,000 of owner withdrawals were made. For the year ended December 31, LoopTech reported a net loss of $38,700. MEEST Prepare a statement of owners' equity for the year ended December 31. If a net loss is incurred or withdrawals were made, enter that amount as a negative number using a minus sign. If an amount box does not require an entry, leave it blank. The following information is available from the records of LoopTech Services. On January 1, Velma Morgan, Capital had a balance of $392,500. During the year, no additional owner investments were made, and $25,000 of owner withdrawals were made. For the year ended December 31, LoopTech reported a net loss of $38,700. MEEST Prepare a statement of owners' equity for the year ended December 31. If a net loss is incurred or withdrawals were made, enter that amount as a negative number using a minus sign. If an amount box does not require an entry, leave it blank.
Expert Answer:
Answer rating: 100% (QA)
Statement of Owners Equity For the Year Ended December 31 ... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these accounting questions
-
1. Prompt the user to enter a number between 5 and 40 inclusive and print the entered number on the screen. If the number is outside the above range, print "out of range". Assumption: User will not...
-
Question 1 This question has two parts, (A) and (B). Answer both parts. 100% Liverpool plc is a company that manufactures a number of different types of electrical goods and has a year end of 31...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
What important retailing decisions to the target market and retailing marketing mix should be considered when investing in: Dunkin Donuts Little Caesars Pizza Starbucks
-
Referring to the previous problem, suppose the rod connecting the two masses m is removed. In this case, the only force between the two masses is their mutual gravitational attraction. In addition,...
-
The lifetime of a certain type of battery is normally distributed with mean value 10 hours and standard deviation 1 hour. There are four batteries in a package. What lifetime value is such that the...
-
How trials may be affected by social media sites?
-
Sales for J. P. Hulett Inc. during the past year amounted to $4 million. Gross profits totaled $1 million, and operating and depreciation expenses were $500,000 and $350,000, respectively. Dividend...
-
QUESTION 3 PART A (30 MARKS) ABC Traders is a business that is a registered VAT vendor. VAT is calculated at 15% where applicable. 1. 2. 3. 4. 56 6. Purchased goods on credit from ABC Traders, R9 200...
-
One technique for limiting fault current is to place reactance in series with the generators. Such reactance can be modeled in PowerWorld Simulator by increasing the value of the generator's positive...
-
The Harris Company started operations this year and reported the following info at the end of the year (i.e ending balances): Equipment. 9800 Notes payable. 12600 Inventory. 5600 Maintenance expenses...
-
what ways can individuals cultivate and demonstrate personal integrity in their professional lives, navigating ethical dilemmas with moral courage, humility, and a steadfast commitment to doing what...
-
The over-50s market has long been ignored by advertising and marketing firms in favour of the market. The complexity of how to appeal to today's mature customers, without targeting their age, has...
-
ABC, Corporation has provided the following data for the month of September: Raw materials purchases..... Direct labor cost $60,000 $27,000 Manufacturing overhead cost ... $76,000 Beginning Ending...
-
On January 1, Year 1, Poultry Processing Company purchased a freezer and related installation equipment for $68,700. The equipment had a three-year estimated life with a $3,300 salvage value....
-
e) If cotx=x lies in second quadrant, find values of other trigonometric functions. 12' f) If a matrix has 8 elements, what are the possible orders it can have.
-
The rocket differential equation in empty space is this: dv(t)M(t)=-udM (t); here M(t) is the rocket mass, at time t, whereas dM(t) is by definition, dM(t)-M(t+dt)-M(t); -dM(1)-1dM() is the mass of...
-
Let (X. A. p) be a measure space. Show that for any A,B A, we have the equality: (AUB)+(An B) = (A) + (B).
-
Clifford Johnson has a limited partnership investment and a rental condominium. Clifford actively manages the rental condominium. During 2012, his share of the loss from the limited partnership was...
-
For each of the following cases, indicate the filing status for the taxpayer(s) for 2012 using the following legend: A - Single B - Married, filing a joint return C - Married, filing separate returns...
-
a. Wilson filed his individual tax return on the original due date, but failed to pay $700 in taxes that were due with the return. If Wilson pays the taxes exactly 2 months late, calculate the amount...
-
When there is a concentration gradient in the system, show that the potential gradient is composed of two terms, (i) an Ohm's-law contribution and (ii) a diffusional contribution. State the equation...
-
Copper is deposited at a cathode from solution with a bulk concentration of \(0.5 \mathrm{M}\) at the rate of \(3.0 \mathrm{~g} / \mathrm{m}^{2} \cdot \mathrm{s}\). Find the surface concentration of...
-
Find the mobility of \(\mathrm{H}^{+}, \mathrm{OH}^{-}\), and other ions from the diffusivity data given in Table 22.1. Table 22.1. Diffusion coefficients of ions in water at 25 C. H+ Na+ K+ Ca+ OH-...

Study smarter with the SolutionInn App


