The following questions are based on the 15 molecules illustrated below. Each molecule may be used...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
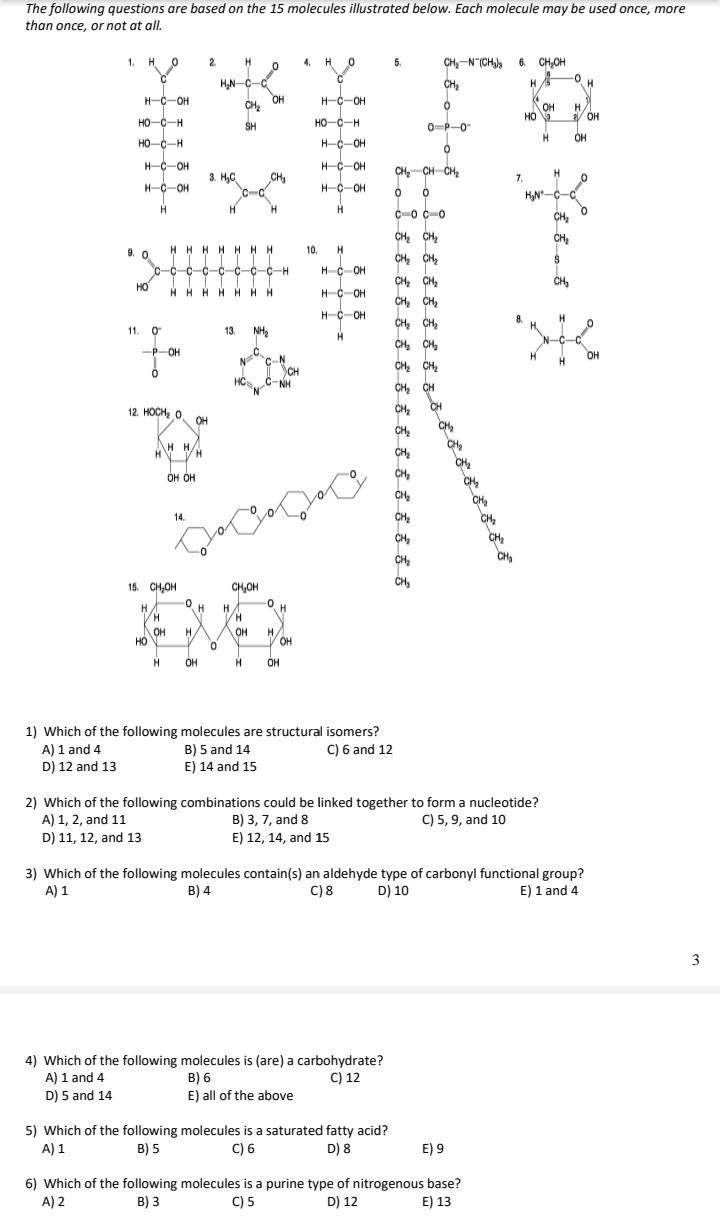
The following questions are based on the 15 molecules illustrated below. Each molecule may be used once, more than once, or not at all. 1. H O 2 4. H O 5. CH,-N (CH, 6 CH,OH CH 0- HN-C-C OH CH H. H-C-OH H-C-OH OH / OH HO-C-H SH HO-C-H 0-P-0 HO-C-H HC-OH H. OH H-C-OH H-C-OH CH, CH CH, 3. H,C CH 7. H-C-OH HC-OH HN-C H H. H. C-o C-0 CH, CH CH2 CH, HHHHHHH 9. 0 c-c-c-c-c-c-c-c-H 10. CH CH2 HC-OH CH CH, CH, CH, CH, CH, CH, HO CH HHHH HHH HC-OH H-C-OH H. 11. 0 13. NH2 CH, CH, CH, CH, CH -P-OH C. N C-N CH C-NH OH 12. HOCH, O CH, CH OH CH, H H/ CH, CH, CH, OH OH CH CH, CH, CH, 14. CH, CH, CH 15. CH,OH CH,OH H. H. H OH HO H. H. V OH OH OH H. OH 1) Which of the following molecules are structural isomers? B) 5 and 14 E) 14 and 15 A) 1 and 4 D) 12 and 13 C) 6 and 12 2) Which of the following combinations could be linked together to form a nucleotide? B) 3, 7, and 8 E) 12, 14, and 15 A) 1, 2, and 11 C) 5, 9, and 10 D) 11, 12, and 13 3) Which of the following molecules contain(s) an aldehyde type of carbonyl functional group? C) 8 A) 1 B) 4 D) 10 E) 1 and 4 4) Which of the following molecules is (are) a carbohydrate? B) 6 E) all of the above C) 12 A) 1 and 4 D) 5 and 14 5) Which of the following molecules is a saturated fatty acid? C) 6 A) 1 B) 5 D) 8 E) 9 6) Which of the following molecules is a purine type of nitrogenous base? C) 5 A) 2 B) 3 D) 12 E) 13 7) Which of the following molecules act as building blocks (monomers) of polypeptides? A) 1, 4, and 6 C) 7, 8, and 13 D) 11, 12, and 13 B) 2, 7, and 8 E) 12, 13, and 15 8) Which of the following molecules is an amino acid with a hydrophobic R group or side chain? A) 3 C) 7 B) 5 D) 8 E) 12 9) Which of the following molecules could be joined together by a peptide bond as a result of a dehydration reaction? A) 2 and 3 B) 3 and 7 C) 7 and 8 D) 8 and 9 E) 12 and 13 10) A fat (or triacylglycerol) would be formed as a result of a dehydration reaction between A) one molecule of 9 and three molecules of 10. B) three molecules of 9 and one molecule of 10. C) one molecule of 5 and three molecules of 9. D) three molecules of 5 and one molecule of 9. E) one molecule of 5 and three molecules of 10. 11) Which of the following molecules could be joined together by a phosphodiester type of covalent bond? A) 3 and 4 D) 11 and 12 C) 6 and 15 B) 3 and 8 E) 11 and 13 12) Which of the following molecules is the pentose sugar found in RNA? A) 1 B) 4 C) 6 D) 12 E) 13 13) Which of the following molecules contains a glycosidic linkage type of covalent bond? A) 4 C) 12 B) 6 D) 13 E) 15 14) Which of the following molecules has (have) a functional group that frequently is involved in maintaining th tertiary structure of a protein? A) 2 B) 3 C) 9 D) 11 E) 9 and 11 15) Which of the following molecules consists of a hydrophilic "head" region and a hydrophobic "tail" region? A) 2 B) 5 C) 7 D) 9 E) 11 16) Which of the following statements is false? A) 1 and 4 could be joined together by a glycosidic linkage to form a disaccharide. B) 9 and 10 could be joined together by ester bonds to form a triacylglycerol. C) 2 and 7 could be joined together to form a short peptide. D) 2, 7, and 8 could be joined together to form a short peptide. E) 14 and 15 could be joined together to form a polypeptide. The following questions are based on the 15 molecules illustrated below. Each molecule may be used once, more than once, or not at all. 1. H O 2 4. H O 5. CH,-N (CH, 6 CH,OH CH 0- HN-C-C OH CH H. H-C-OH H-C-OH OH / OH HO-C-H SH HO-C-H 0-P-0 HO-C-H HC-OH H. OH H-C-OH H-C-OH CH, CH CH, 3. H,C CH 7. H-C-OH HC-OH HN-C H H. H. C-o C-0 CH, CH CH2 CH, HHHHHHH 9. 0 c-c-c-c-c-c-c-c-H 10. CH CH2 HC-OH CH CH, CH, CH, CH, CH, CH, HO CH HHHH HHH HC-OH H-C-OH H. 11. 0 13. NH2 CH, CH, CH, CH, CH -P-OH C. N C-N CH C-NH OH 12. HOCH, O CH, CH OH CH, H H/ CH, CH, CH, OH OH CH CH, CH, CH, 14. CH, CH, CH 15. CH,OH CH,OH H. H. H OH HO H. H. V OH OH OH H. OH 1) Which of the following molecules are structural isomers? B) 5 and 14 E) 14 and 15 A) 1 and 4 D) 12 and 13 C) 6 and 12 2) Which of the following combinations could be linked together to form a nucleotide? B) 3, 7, and 8 E) 12, 14, and 15 A) 1, 2, and 11 C) 5, 9, and 10 D) 11, 12, and 13 3) Which of the following molecules contain(s) an aldehyde type of carbonyl functional group? C) 8 A) 1 B) 4 D) 10 E) 1 and 4 4) Which of the following molecules is (are) a carbohydrate? B) 6 E) all of the above C) 12 A) 1 and 4 D) 5 and 14 5) Which of the following molecules is a saturated fatty acid? C) 6 A) 1 B) 5 D) 8 E) 9 6) Which of the following molecules is a purine type of nitrogenous base? C) 5 A) 2 B) 3 D) 12 E) 13 7) Which of the following molecules act as building blocks (monomers) of polypeptides? A) 1, 4, and 6 C) 7, 8, and 13 D) 11, 12, and 13 B) 2, 7, and 8 E) 12, 13, and 15 8) Which of the following molecules is an amino acid with a hydrophobic R group or side chain? A) 3 C) 7 B) 5 D) 8 E) 12 9) Which of the following molecules could be joined together by a peptide bond as a result of a dehydration reaction? A) 2 and 3 B) 3 and 7 C) 7 and 8 D) 8 and 9 E) 12 and 13 10) A fat (or triacylglycerol) would be formed as a result of a dehydration reaction between A) one molecule of 9 and three molecules of 10. B) three molecules of 9 and one molecule of 10. C) one molecule of 5 and three molecules of 9. D) three molecules of 5 and one molecule of 9. E) one molecule of 5 and three molecules of 10. 11) Which of the following molecules could be joined together by a phosphodiester type of covalent bond? A) 3 and 4 D) 11 and 12 C) 6 and 15 B) 3 and 8 E) 11 and 13 12) Which of the following molecules is the pentose sugar found in RNA? A) 1 B) 4 C) 6 D) 12 E) 13 13) Which of the following molecules contains a glycosidic linkage type of covalent bond? A) 4 C) 12 B) 6 D) 13 E) 15 14) Which of the following molecules has (have) a functional group that frequently is involved in maintaining th tertiary structure of a protein? A) 2 B) 3 C) 9 D) 11 E) 9 and 11 15) Which of the following molecules consists of a hydrophilic "head" region and a hydrophobic "tail" region? A) 2 B) 5 C) 7 D) 9 E) 11 16) Which of the following statements is false? A) 1 and 4 could be joined together by a glycosidic linkage to form a disaccharide. B) 9 and 10 could be joined together by ester bonds to form a triacylglycerol. C) 2 and 7 could be joined together to form a short peptide. D) 2, 7, and 8 could be joined together to form a short peptide. E) 14 and 15 could be joined together to form a polypeptide.
Expert Answer:
Answer rating: 100% (QA)
7 Molecule 4is the structure of D Glucose and CA Epimor change at a Hace C4 at... View the full answer

Related Book For 

Intermediate accounting
ISBN: 978-0077647094
7th edition
Authors: J. David Spiceland, James Sepe, Mark Nelson
Posted Date:
Students also viewed these chemical engineering questions
-
The following questions are adapted from a variety of sources including questions developed by the AICPA Board of Examiners and those used in the Kaplan CPA Review Course to study property, plant,...
-
The following questions are adapted from a variety of sources including questions developed by the AICPA Board of Examiners and those used in the Kaplan CPA Review Course to study property, plant,...
-
The following questions are part of an internal control questionnaire for the acquisition and expenditure process. The client has been asked to answer the questions for the acquisition and...
-
Problem 5-47 Amortizing Loans and Inflation (LO3) Suppose you take out a $108,000, 20-year mortgage loan to buy a condo. The interest rate on the loan is 5%. To keep things simple, we will assume you...
-
Okanagan Tire Corporation redesigned its production facilities around a team-based system. However, the company president believes that employees will not be motivated unless they receive incentives...
-
Define method overloading.
-
Christopher Boling was seriously injured in 2008 when vapors escaping from a gas can ignited. He filed a products liability claim against the manufacturer. To fund the litigation, Boling entered into...
-
The Mobile Oil company has recently acquired oil rights to a new potential source of natural oil in Alaska. The current market value of these rights is $90,000. However, if there is natural oil at...
-
Discuss the business benefits of adopting the relational database model within an organization. How does a relational model create or enhance business value? Are there any limitations to using a...
-
To enhance heat transfer from a silicon chip of width W = 4 mm on a side, a copper pin fin is brazed to the surface of the chip. The pin length and diameter are L = 12 mm and D = 2 mm, respectively,...
-
What are some of the measures that can be taken to ensure that data in a cloud environment is secure?
-
C Kevin is having financial problems. He has not been receiving funds from some accounts receivable according to the trading terms and also has not been paying some credit suppliers according to...
-
Having checked the monthly accounts of Honey Suckle for account allocations, three errors were found that needed correction through the general journal. Subsidiary ledgers and control accounts are...
-
Show the following transactions in the two journals of S Alderson. Assume first that periodic inventory applies and then that perpetual inventory applies. 19 June 2022: Goods were sold to R...
-
Inventory was delivered to a customer on 30 June 2022 valued at $7579 ($6890 + $689 GST). However, there was a problem with the invoicing process and a tax invoice was not raised for the customer....
-
Show the following transactions in the two journals of P Kulevska. Assume that perpetual inventory applies. 5 October 2022: P Kulevska deposited $792 into her bank account. This was from a cash sale...
-
If a firm has a Return on Assets higher than the industry average, while its Return on Equity is below the industry average, what must be true about the firm? A. It has a higher total asset turnover...
-
A heat engine has a heat input of 3 Ã 104 Btu/h and a thermal efficiency of 40 percent. Calculate the power it will produce, in hp. Source 3 x 10 Btu/h 40% HE Sink
-
On January 1, 2013, Burleson Corporations projected benefit obligation was $30 million. During 2013 pension benefits paid by the trustee were $4 million. Service cost for 2013 is $12 million. Pension...
-
At January 1, 2013, NCI Industries, Inc., was indebted to First Federal Bank under a $240,000, 10% unsecured note. The note was signed January 1, 2009, and was due December 31, 2014. Annual interest...
-
On January 1, Seneca Asset Management enters into a contract with a client to provide fund management services for one year. The client is required to pay a fixed amount of $100,000 at the end of...
-
What type of accounts are accounts receivable and inventory? (a) Cash accounts. (b) Operating accounts. (c) Financing accounts. (d) Investing accounts.
-
Which of the following is a cash outflow? (a) Proceeds from borrowing. (b) Repayments of debt principal. (c) Payment for taxes. (d) Both (b) and (c).
-
How would payments for taxes be classified? (a) Operating outflow. (b) Operating inflow. (c) Investing outflow. (d) Financing outflow

Study smarter with the SolutionInn App


