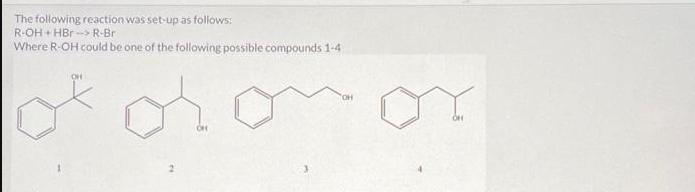
The following reaction was set-up as follows: R-OH HBr R-Br Where R-OH could be one of...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
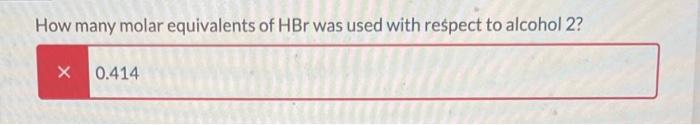
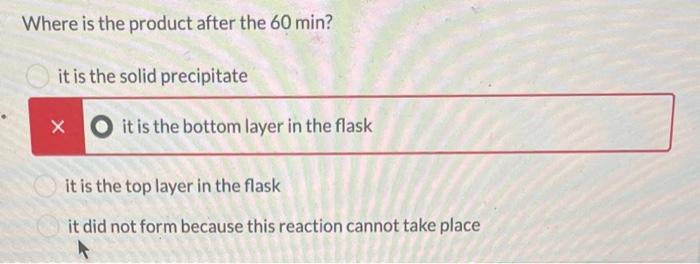
The following reaction was set-up as follows: R-OH HBr R-Br Where R-OH could be one of the following possible compounds 1-4 5.0 mL of the unknown alcohol was added to 15 mL of HBr ( 48% HBr in water) in a 150 mL Erlenmeyer flask. The reaction was covered and stirred at room temperature for 60 min. After that time, two layers could be seen in the flask. One was clear and the other was a pale yellow. The following table has the physical properties for each of the possible alcohols and HBr. All of the alcohols are immiscible with water. Compound 1 2 3 4 HBr (48%) Molar mass Melting point (g/mol) (degC) 136.19 28-23 136.19 136.19 136.19 80.91 -18 -- Boiling point (degC) 202 110-111 119-121 219-221 -- Density (g/mL at 25 C) 0.973 0.975 1.001 0.973 1.49 What needs to be done to balance the equation shown? Add H2O to the reactants side X Remove H2O from the reactants side Add H2O to the products side Remove H2O from the products side How many molar equivalents of HBr was used with respect to alcohol 2? X 0.414 Where is the product after the 60 min? it is the solid precipitate X it is the bottom layer in the flask it is the top layer in the flask it did not form because this reaction cannot take place 13 2 points How would the product and the starting alcohol differ in IR? Both have aromatic C-H bonds but only the product will have C=C aromatic bonds in the IR The product should have sp3 C-H bonds and the alcohol will not The product should not have an O-H bond, but will still have the sp3 and aromatic C-H bonds The product should not have an O-H bond or the sp3 C-H bonds The following reaction was set-up as follows: R-OH HBr R-Br Where R-OH could be one of the following possible compounds 1-4 5.0 mL of the unknown alcohol was added to 15 mL of HBr ( 48% HBr in water) in a 150 mL Erlenmeyer flask. The reaction was covered and stirred at room temperature for 60 min. After that time, two layers could be seen in the flask. One was clear and the other was a pale yellow. The following table has the physical properties for each of the possible alcohols and HBr. All of the alcohols are immiscible with water. Compound 1 2 3 4 HBr (48%) Molar mass Melting point (g/mol) (degC) 136.19 28-23 136.19 136.19 136.19 80.91 -18 -- Boiling point (degC) 202 110-111 119-121 219-221 -- Density (g/mL at 25 C) 0.973 0.975 1.001 0.973 1.49 What needs to be done to balance the equation shown? Add H2O to the reactants side X Remove H2O from the reactants side Add H2O to the products side Remove H2O from the products side How many molar equivalents of HBr was used with respect to alcohol 2? X 0.414 Where is the product after the 60 min? it is the solid precipitate X it is the bottom layer in the flask it is the top layer in the flask it did not form because this reaction cannot take place 13 2 points How would the product and the starting alcohol differ in IR? Both have aromatic C-H bonds but only the product will have C=C aromatic bonds in the IR The product should have sp3 C-H bonds and the alcohol will not The product should not have an O-H bond, but will still have the sp3 and aromatic C-H bonds The product should not have an O-H bond or the sp3 C-H bonds
Expert Answer:
Answer rating: 100% (QA)
Introduction Pressure loss in process pipelines often referred to as pressure drop is a phenomenon that occurs as a fluid flows through a pipeline system It is a critical aspect in the design and oper... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
123 Compare the purely graphical properties of these two notations, and the ways in which the graphical properties of each display correspond to the information structure being defined. Describe...
-
Selected data from a February payroll register for Halverson Company are presented below. Some amounts are intentionally omitted. FICA taxes are 7.65%. State income taxes are 4% of gross earnings....
-
To operate a given flash lamp requires a charge of 38 (C. What capacitance is needed to store this much charge in a capacitor with a potential difference between its plates of 9.0 V?
-
At the end of July, Tony took a complete inventory of his supplies and found the following: 5 dozen screws at a cost of $8 a dozen 2 dozen screws at a cost of $5 a dozen 2 cartons of computer...
-
In a contract dispute between a US company and a Canadian company, the contract itself referred to provisions of the Uniform Commercial Code. Do these references alone preempt the contract from being...
-
Your boss at Scott Sampson Products, Inc., has just provided you with the schedule and lead times for the bracket in Problem 14.8. The unit is to be prepared in week 10. The lead times for the...
-
Problem 5. [20 pts] Verify Green's Theorem for the following vector field and region D. Namely, compute both, the line integral and the double integral. x1 1. (10pts) F (1,-1). and D is the square...
-
Using resources from the Internet, research the issue of collection of sales and use tax on internet sales. Your Research should include a discussion of the U.S. Supreme Court opinion in South Dakota...
-
Warrenia has two regions. In Oliviland, the marginal benefit associated with pollution cleanup is MB = 300 10Q, while in Linneland, the marginal benefit associated with pollution cleanup is MB = 200...
-
The private marginal benefit associated with a products consumption is PMB = 350 4Q and the private marginal cost associated with its production is PMC = 6Q. Furthermore, the marginal external...
-
There is concern that Californias cap-and-trade system for greenhouse gases, implemented in 2012, has led to hot spots of pollutionlocalized areas with very high concentrations of hazardous...
-
Firms A and B each produce 80 units of pollution. The federal government wants to reduce pollution levels. The marginal costs associated with pollution reduction are MC A = 50 + 3Q A for firm A and...
-
Two firms are ordered by the federal government to reduce their pollution levels. Firm As marginal costs associated with pollution reduction is MC = 150 + 3Q. Firm Bs marginal costs associated with...
-
Ms. Snow owns a city home in Hope, BC as well as a chalet in a local ski area. The city home was purchased in 2013 at a cost of $188,000. The chalet was purchased in 2006 for $89,000. On July 1,...
-
Juanita owns a home in Richardson, TX. She purchases a Homeowners Policy (HO-3) from Farm State Ins. Co. The policy provides $100,000 in liability coverage (coverage E) and $5,000 in Med Pay coverage...
-
Consider the following reaction: If the equilibrium partial pressures of N2, O2, and NO are 0.15 atm, 0.33 atm, and 0.050 atm, respectively, at 2200°C, what is KP? N2(g) + O2(g) = 2NO(g)
-
The formula for calculating the energies of an electron in a hydrogen-like ion is given in Problem 8.57. This equation cannot be applied to many-electron atoms. One way to modify it for the more...
-
(a) The formula of the simplest hydrocarbon is CH4 (methane). Predict the formulas of the simplest compounds formed between hydrogen and the following elements: silicon, germanium, tin, and lead. (b)...
-
Resources should be estimated against which entity: a. The deliverables level of the WBS b. Critical project constraints c. The Risk Breakdown Structure d. The schedule activities
-
Resource leveling is a method to: a. Make sure everyone working on the project has approximately the same amount of work to accomplish b. A technique in which start and finish dates are adjusted...
-
Your customer has requested a minor change to the project that shouldnt impact the schedule. How should you respond? a. Authorize the change to maintain good customer relations b. Refuse the change...

Study smarter with the SolutionInn App


