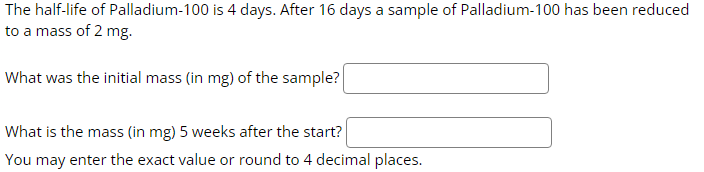
The half-life of Palladium-100 is 4 days. After 16 days a sample of Palladium-100 has been...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The half-life of Palladium-100 is 4 days. After 16 days a sample of Palladium-100 has been reduced to a mass of 2 mg. What was the initial mass (in mg) of the sample? What is the mass (in mg) 5 weeks after the start? You may enter the exact value or round to 4 decimal places. The half-life of Palladium-100 is 4 days. After 16 days a sample of Palladium-100 has been reduced to a mass of 2 mg. What was the initial mass (in mg) of the sample? What is the mass (in mg) 5 weeks after the start? You may enter the exact value or round to 4 decimal places.
Expert Answer:
Answer rating: 100% (QA)
Palladium100 Decay Initial Mass Since the halflife is 4 days and the mass has been reduced to 2 mg a... View the full answer

Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
The risk hee rate is 1 99% and the market risk premium is 5 98% A stock whap of 96 just paid a dividend of $123 The dividend is expected to grow at 21.37% for five years and then grow at 4 75%...
-
You are required to write a Python program that will manage character (heroes and villain) information. Character (hero and villain) information will be stored in a text file that will be read in...
-
In Exercises 7582, express the given function h as a composition of two functions f and g so that h(x) = (f g)(x). h(x) = x-9
-
Suppose a fair coin is tossed 6 times. What is the probability that (a) 6 heads will occur? (b) 3 heads will occur? (c) 2 heads will occur?
-
An astronaut in orbit can just resolve two point sources on the earth that are 75.0 m apart. Assume that the resolution is diffraction limited, and use Rayleigh's criterion. What is the astronaut's...
-
Simulate tossing a coin. (a) For a balanced coin, generate 100 flips. (b) For a coin with probability of heads 0.8, generate 100 flips.
-
Filer manufacturing has 8.3 million shares of common stock outstanding. The current share price is $53 and the book value per share is $4. Filer manufacturing also has two bond issues outstanding....
-
(a) Table 1 shows a truth table. Table 1 A 0 BO C X 0 0 1 0 0 1 1 0 1 0 1 0 1 1 1 1 0 0 1 1 0 1 0 1 1 0 1 1 1 1 0 (i) Produce a Boolean expression for X in terms of A, B and C based on the truth...
-
The proposed rates were not in the range the CEO expected given the pricing analysis. The CEO has asked the pricing actuary to verify the total projected loss cost excluding potential large storm...
-
Number 11.20 and 11.21. Please help me eling north at 990 km/h 11. at a latitude of 60 north. 11.20. A particle of mass m is dropped from rest at a height of 500 m. Where does it hit the ground?...
-
Is any magnetic field produced when (a) a uniformly charged sphere rotates at constant rotational speed about an axis that passes through its fixed center, \((b)\) a charged particle moves...
-
By focusing an ultraviolet laser pulse, you can produce an apparently hovering plasma point in midair. If it takes an electric field magnitude of \(1.0 \times 10^{6} \mathrm{~N} / \mathrm{C}\) to...
-
It takes sunlight about \(8 \mathrm{~min}\) to travel from the Sun to Earth, where it has an average intensity of \(1400 \mathrm{~W} / \mathrm{m}^{2}\). If it takes \(44 \mathrm{~min}\) for light to...
-
A parallel-plate capacitor with circular plates has a steady charging current of \(5.0 \mathrm{~A}\). The wires into and out of the plates attach to the plate centers. If the radius of each plate is...
-
Assume that you have shorted the call option in Problem 2. a. If the stock is trading at $46 in three months, what will you owe? b. If the stock is trading at $32 in three months, what will you owe?...
-
Proprietor: Ms. Nicole Coffeur Name: Description: Hair and Beauty Salon Teenage Style Bag The business has the following opening balances: Cash At Bank Account Receivable Motor Vehicle Account...
-
Let (x) = x 2 - 9, g(x) = 2x, and h(x) = x - 3. Find each of the following. (((--) 2
-
The following transactions pertain to the March 31, 2013, year end, for a province reporting in accordance with the CICA PSA Handbook: 1. Assume the accrual for the year-end estimate of income tax...
-
Esson Oil buys oil at market prices and sells it at market price plus a profit margin of U.S. $5 per barrel. On January 1, Esson buys 10 barrels of oil at a price of U.S. $30 per barrel when the...
-
On January 1, 2013, Sienna acquired all the shares of Danon for $160,000. The financial statements of the two entities at December 31, 2013, contained the following information: Additional...
-
How many structural isomers are there for hydrocarbons that have the molecular formula C 4 H 10 ? (a) none (b) one (c) two (d) three
-
Which contains more hydrogen atomsa five-carbon saturated hydrocarbon molecule or a five-carbon unsaturated hydrocarbon molecule? (a) The unsaturated hydrocarbon has more hydrogen atoms. (b) The...
-
One solution to the problem of our overflowing landfills is to burn plastic objects instead of burying them. What are some advantages and disadvantages of this practice? (a) disadvantage: toxic air...

Study smarter with the SolutionInn App


