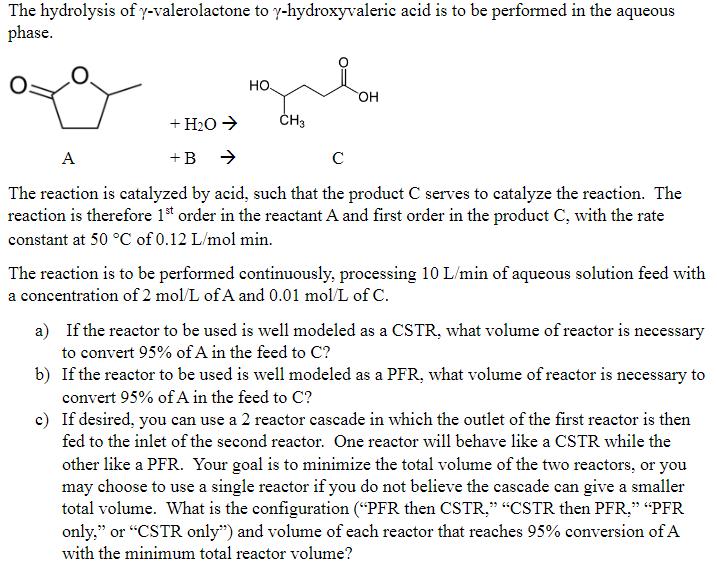
The hydrolysis of y-valerolactone to y-hydroxyvaleric acid is to be performed in the aqueous phase. +...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The hydrolysis of y-valerolactone to y-hydroxyvaleric acid is to be performed in the aqueous phase. + HO +B HO. CH3 OH A The reaction is catalyzed by acid, such that the product C serves to catalyze the reaction. The reaction is therefore 1st order in the reactant A and first order in the product C, with the rate constant at 50 C of 0.12 L/mol min. The reaction is to be performed continuously, processing 10 L/min of aqueous solution feed with a concentration of 2 mol/L of A and 0.01 mol/L of C. a) If the reactor to be used is well modeled as a CSTR, what volume of reactor is necessary to convert 95% of A in the feed to C? b) If the reactor to be used is well modeled as a PFR, what volume of reactor is necessary to convert 95% of A in the feed to C? c) If desired, you can use a 2 reactor cascade in which the outlet of the first reactor is then fed to the inlet of the second reactor. One reactor will behave like a CSTR while the other like a PFR. Your goal is to minimize the total volume of the two reactors, or you may choose to use a single reactor if you do not believe the cascade can give a smaller total volume. What is the configuration ("PFR then CSTR." "CSTR then PFR," "PFR only," or "CSTR only") and volume of each reactor that reaches 95% conversion of A with the minimum total reactor volume? The hydrolysis of y-valerolactone to y-hydroxyvaleric acid is to be performed in the aqueous phase. + HO +B HO. CH3 OH A The reaction is catalyzed by acid, such that the product C serves to catalyze the reaction. The reaction is therefore 1st order in the reactant A and first order in the product C, with the rate constant at 50 C of 0.12 L/mol min. The reaction is to be performed continuously, processing 10 L/min of aqueous solution feed with a concentration of 2 mol/L of A and 0.01 mol/L of C. a) If the reactor to be used is well modeled as a CSTR, what volume of reactor is necessary to convert 95% of A in the feed to C? b) If the reactor to be used is well modeled as a PFR, what volume of reactor is necessary to convert 95% of A in the feed to C? c) If desired, you can use a 2 reactor cascade in which the outlet of the first reactor is then fed to the inlet of the second reactor. One reactor will behave like a CSTR while the other like a PFR. Your goal is to minimize the total volume of the two reactors, or you may choose to use a single reactor if you do not believe the cascade can give a smaller total volume. What is the configuration ("PFR then CSTR." "CSTR then PFR," "PFR only," or "CSTR only") and volume of each reactor that reaches 95% conversion of A with the minimum total reactor volume?
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
What are some techniques of good writers? Which ones do you use regularly?
-
Is this an ethical approach?
-
Sandra Martins, a confectionery based in Austria, decides to produce peanut packs for supermarkets in Salzburg. The projected sales units and selling price per pack for 2021 for its three brands of...
-
A baseball is thrown by a pitcher at \(95 \mathrm{mph}\) through standard air. The diameter of the baseball is \(2.82 \mathrm{in}\). Estimate the drag force on the baseball. Explain how the actual...
-
Given the following data, calculate: (a) BEPx; (b) BEP$; (c) The profit at 100,000 units: P = $8/unit V = $4/unit F = $50,000
-
From experiences as a hospital laboratory manager in Fiji, discuss strategies that you used or was already in placed in your organization to maintain effective relationship. Highlight the...
-
A plant is already 11.00 meters tall, and it will grow 5 centimeters every month. The plant's height, H (in meters ), after x months is given by the following function. H(x)=11.00+0.05x What is the p...
-
NASA plans to use \(x\)-ray diffraction to identify minerals on the surface of Mars. If the wavelength of the \(x\) rays is \(0.155 \mathrm{~nm}\), what is the lattice spacing in a crystalline sample...
-
Babinet's principle states that, except for differences in intensity, the interference pattern created by light passing around an opaque object is the same as the pattern created by the same light...
-
Consider 500 uniform electric field lines that pass through a hollow tube of length \(10 \mathrm{~m}\) and cross-sectional area \(1 \mathrm{~m}^{2}\). If the tube is cut at the middle in such way the...
-
An electron is fired toward a nonconducting infinite sheet carrying a uniformly distributed charge from 5.00 \(\mathrm{m}\) away. The electron has an initial speed of \(3 \times 10^{6} \mathrm{~m} /...
-
Let X X be a BES 3 3 starting from 0 . Prove that 1 / X 1 / X is a local martingale, but not a martingale. Establish that, for u < 1 u < 1 , E ( 1 X 1 R u ) = 1 X u 2 ( X u 1 u ) E ( 1 X 1 | R...
-
Jim Anderson is a training specialist in the human resource department of a large pharmaceutical company. In response to a recent company wide survey, Jim specifically designed a 6-week training...
-
Wilsons Auto Repair ended 2011 with Accounts Receivable of $85,000 and a credit balance in Allowance for Uncollectible Accounts balance of $11,000. During 2012, Wilsons Auto Repair had the following...
-
Can you name the English rock group that was famous worldwide in the 1970s and 1980s and whose lead singer was Freddie Mercury? The group had a resurgence in 2019 when Adam Lambert took over for...
-
How would you modify Table 6-2 for a. A constant-volume gas-phase reaction, and b. A variable-volume gas-phase reaction? Table 6-2 1. Mole balances: BR dNAdt=rAV dNBdt=rBV dNCdt=rCV dNDdt=rDV PFR PBR...
-
Ethyl acetate is an extensively used solvent and can be formed by the vapor-phase esterification of acetic acid and ethanol. The chemical equation shows the formation of Ethyl acetate by the vapor...
-
In 2022, Mark purchased two separate activities. Information regarding these activities for 2022 and 2023 is as follows: The 2022 losses were suspended losses for that year. During 2023, Mark also...
-
In 2023, Julie, a single individual, reported the following items of income and deduction: Julie owns 100% and is an active participant in the rental real estate activity. What is her taxable income...
-
Jerry sprayed all of the landscaping around his business with a pesticide in June 2023. Shortly thereafter, all of the trees and shrubs unaccountably died. The FMV and the adjusted basis of the...

Study smarter with the SolutionInn App


