The irreversible first-order (wrt partial pressure of A) gas-phase reaction: A B is carried out...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
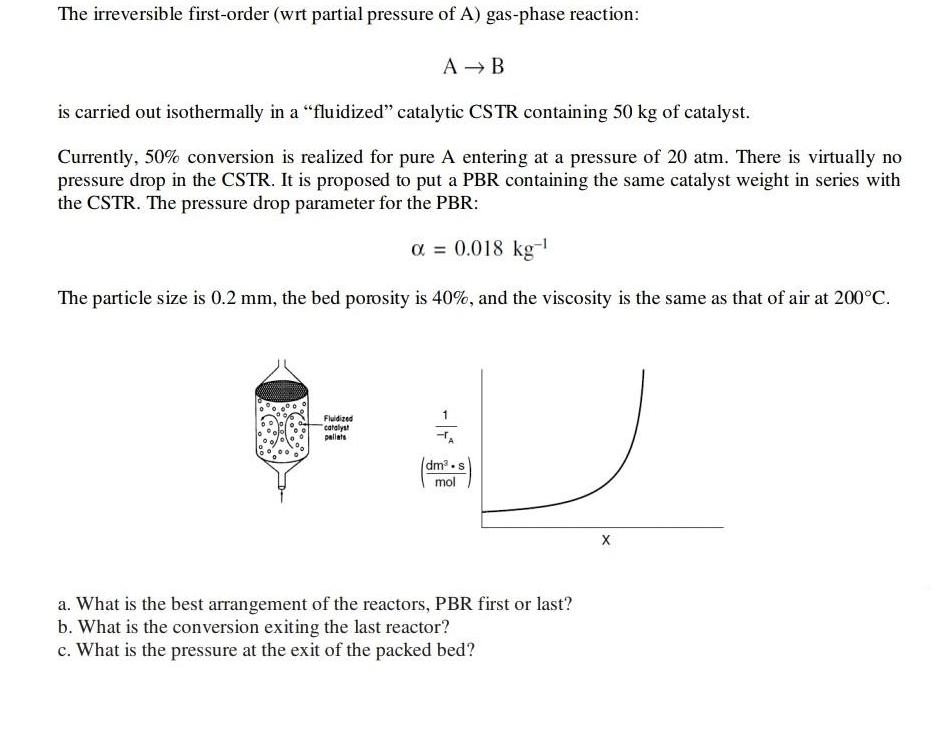
The irreversible first-order (wrt partial pressure of A) gas-phase reaction: A → B is carried out isothermally in a "fluidized" catalytic CSTR containing 50 kg of catalyst. Currently, 50% conversion is realized for pure A entering at a pressure of 20 atm. There is virtually no pressure drop in the CSTR. It is proposed to put a PBR containing the same catalyst weight in series with the CSTR. The pressure drop parameter for the PBR: α = 0.018 kg-¹ The particle size is 0.2 mm, the bed porosity is 40%, and the viscosity is the same as that of air at 200°C. Fluidized catalyst pallats -TA dm³-s mol X a. What is the best arrangement of the reactors, PBR first or last? b. What is the conversion exiting the last reactor? c. What is the pressure at the exit of the packed bed? The irreversible first-order (wrt partial pressure of A) gas-phase reaction: A → B is carried out isothermally in a "fluidized" catalytic CSTR containing 50 kg of catalyst. Currently, 50% conversion is realized for pure A entering at a pressure of 20 atm. There is virtually no pressure drop in the CSTR. It is proposed to put a PBR containing the same catalyst weight in series with the CSTR. The pressure drop parameter for the PBR: α = 0.018 kg-¹ The particle size is 0.2 mm, the bed porosity is 40%, and the viscosity is the same as that of air at 200°C. Fluidized catalyst pallats -TA dm³-s mol X a. What is the best arrangement of the reactors, PBR first or last? b. What is the conversion exiting the last reactor? c. What is the pressure at the exit of the packed bed?
Expert Answer:
Answer rating: 100% (QA)
Step 1 of 4 a The irreversible gasphase reaction is as follows A B The re... View the full answer

Related Book For 

Introduction to Chemical Engineering Thermodynamics
ISBN: 978-0071247085
7th edition
Authors: J. M. Smith, H. C. Van Ness, M. M. Abbott
Posted Date:
Students also viewed these physics questions
-
The irreversible first-order (wrt partial pressure of A) gas-phase reaction A B is carried out isothermally in a "fluidized" catalytic CSTR containing 50 kg of catalyst. Currently, 50% conversion is...
-
The production of maleic anhydride by the air oxidation of benzene was studied using a vanadium pentoxide catalyst [Chem Eng Sci, 43, 1051 (1988)]. The reactions that occur are C6H6 + 9/ 2 O2 C4H2O3...
-
1) A gas phase reaction A B takes place in a packed bed reactor. The pressure gradient arised along the length of PBR is assumed to be constant as dP / dW = -0.2 atm.kg. The conversion achieved in...
-
What are the lessons of the 1997 Asian financial crisis? Were the IMF supported adjustment programs successful in their objective? Were there flaws in their design?
-
The management of Heckel Communications Inc. is considering two capital investment projects. The estimated net cash flows from each project are as follows: The radio station requires an investment of...
-
List and briefly explain two common ways of generating the condition to be tested in a conditional branch instruction.
-
The coefficient of determination R2 measures the percentage of variation in the outcome that is explained by the model. In Exercises 9 and 10, determine whether the statement is true or false. If the...
-
Consider the following transactional data for the first month of operations for Shine King Cleaning. Nov. 1 Stockholders contributed $ 35,000 and a truck, with a market value of $ 8,000, to the...
-
You have read the Case Study, worked through the examples and exercises embedded in it, and had a chance to reflect on some of the complexities of running this business. Post your replies to the...
-
During 2020, you were hired as the Chief Financial Officer for MC Travel Inc., a fairly young travel company that is growing quickly. A key accounting staff member has prepared the financial...
-
It was a hot summer day in May 2016 when Li An, the president of the Pakistani branch of the China Guangxi Corporation for International Techno-Economic Cooperation (GCITEC), received a startling...
-
Break into two or three groups to research the use of affinity cards. First, develop a list of affinity cards and their sponsors. Does your university sponsor a card? Next, each group should choose a...
-
Read your parents' life insurance policy, or that of a friend or relative, cover to cover. Prepare a one-page report of key policy features including beneficiary designation, policy clauses,...
-
Interview a friend employed in your career field or interview a benefits representative from a company agency in your career field. Discuss the benefits that you might expect to receive as an...
-
The draft accounts for the year ended 30 June 2019 and a balance sheet as at that date for S. Steiner are submitted to you. Towards the end of the financial year her accountant resigned and she...
-
As a part of a group project, interview a representative from a nursing home, extended care facility, or other assisted living facility and discuss the costs and payment methods used. Are residents...
-
Identify the intervention strategies that managers can use to change the culture in their organizations. Avoiding the socialization of new members of an organization Removing people who deviate from...
-
Derive Eq. (18.33) from Eq. (18.32).
-
A stream of propane gas is partially liquefied by throttling from 200 bar and 370 K to 1 bar. What fraction of the gas is liquefied in this process? The vapor pressure of propane is given by Kq....
-
Assuming the validity of the De Priester charts, make the following VLE calculations far the ethane (1)/propane(2)/isobutane(3)/isopentane(4) system: (a) BVBL P, given x1 = 0.10, x2 = 0.20. x3 =...
-
Wet steam at 1,100 kPa expands at constant enthalpy (as in a throttling process) to 101.33 kPa, where its temperature is 105oC. What is the quality' of the steam in its initial state?
-
How does an auditor make a preliminary judgment about materiality during the planning phase?
-
In the Hochfelder case all of the following were factors in the case except a. The Securities Exchange Act of 1934. Data From Exchange Act 1934 When the Securities Act was passed, the Interstate...
-
Refer to the example of an auditors report issued in 1915. List the differences between the report styles in 1915 and today (refer to Chapter 1). Indicate in what ways this report would be deficient...

Study smarter with the SolutionInn App


