At midnight, a hospital emergency room (E.R.) has 3 patients. The rate that new patients are...
Fantastic news! We've Found the answer you've been seeking!
Question:

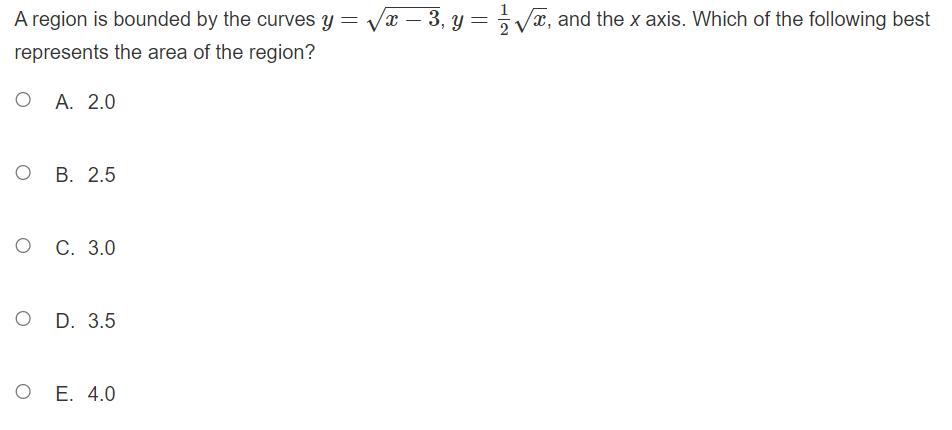
Transcribed Image Text:
At midnight, a hospital emergency room (E.R.) has 3 patients. The rate that new patients are admitted to the E.R. over the next 24 hours is modeled by the function A (t) =(768 +23t-t²). The rate that E.R. patients are released over the same time period is modeled by the function 65 R (t) = (390+41t – t²). The units for both A (t) and R (t) are in people per hour. Approximately how many patients are in the E.R. at the end of the 24-hour time period? OA. 3 O B. 5 O C. 7 O D. 9 OE. 11 A region is bounded by the curves y = √√x - 3, y = √, and the x axis. Which of the following best represents the area of the region? OA. 2.0 O B. 2.5 O C. 3.0 D. 3.5 OE. 4.0 At midnight, a hospital emergency room (E.R.) has 3 patients. The rate that new patients are admitted to the E.R. over the next 24 hours is modeled by the function A (t) =(768 +23t-t²). The rate that E.R. patients are released over the same time period is modeled by the function 65 R (t) = (390+41t – t²). The units for both A (t) and R (t) are in people per hour. Approximately how many patients are in the E.R. at the end of the 24-hour time period? OA. 3 O B. 5 O C. 7 O D. 9 OE. 11 A region is bounded by the curves y = √√x - 3, y = √, and the x axis. Which of the following best represents the area of the region? OA. 2.0 O B. 2.5 O C. 3.0 D. 3.5 OE. 4.0
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these mathematics questions
-
There is only one value of the equilibrium constant for a particular system at a particular temperature, but there are an infinite number of equilibrium positions. Explain.
-
Calculate the KP for this reaction at 298 K if the Keq = 1.76 103. 3O2 (g) + 2O3(g)
-
Calculate the KP for this reaction at 310 K if the Keq = 6.22 103. 4NO2 (g) + O2 (g) + 2N2O5 (g)
-
Foreman Publishing Companys income for the most recent quarter was $500,000, and the average net book value of assets during the quarter was $1.5 million. If the company has a required rate of return...
-
Consider hydrogen cyanide, H C N. (a) What is the hybridization at the N at the C? (b) What are the types of the three CN bonds? What orbitals overlapping to form them? (c) In what type of orbital...
-
Computech Corporation is expanding rapidly and currently needs to retain all of its earnings; hence, it does not pay dividends. However, investors expect Computech to begin paying dividends,...
-
Assume that the British pounds 1-year forward rate exhibits a discount. Assume that interest rate parity continually exists. Explain how the discount on the British pounds 1-year forward discount...
-
Portions of the 20X2 financial statements of Williams Company, a paint manufacturer, are reproduced below (in thousands of dollars): Partial Income Statement for the year ended 31 December 20X2 Net...
-
Brief Exercise 4-13 (Algo) Record employee purchases (LO4-6) Clooney Corporation establishes a petty cash fund for $200 and issues a credit card to its office manager. By the end of the month,...
-
The dean of a school of nursing (SON) and the associate deans of undergraduate and graduate programs have experienced increasing frustration with a lack of clinical placements for the students. The...
-
Urban oriented development programmes generated urban centred employment which resulted into (A) Rural to Rural Migration (B) Urban to Rural Migration (C) Rural to Urban Migration (D) Urban to Urban...
-
Joe sold stock in the following amounts during the current tax year ( 2 0 2 2 ) . Purchase: 4 5 , 0 0 0 Sale: 2 5 , 0 0 0 Purchase: 1 / 1 / 2 0 2 2 Sale: 7 / 1 / 2 0 2 2 Purchase: 4 0 , 0 0 0 Sale:...
-
Korkdam Breweries Limited (KBL), a major beer manufacturer takes prides in its excellent performance. Tracking performance from plant to the warehouse, the Managing Director; Jack Singaro was pleased...
-
find at least two recent articles that describes new or developing technology that will have significant influence on supply chain management within the next 5-6 years. You will need to add your...
-
Consider a double-paned window consisting of two panes of glass, each with a thickness of 0.600 cm and an area of 0.700 m separated by a layer of air with a thickness of 1.90 cm. The temperature on...
-
1.1 Introduction: State the objective i.e. why your help is needed. State the project goal, plus any constraints of conditions. Breakdown the goal into several general services your Company will...
-
The direct labour budget of Yuvwell Corporation for the upcomingfiscal year contains the following details concerning budgeteddirect labour-hours:1st Quarter2nd Quarter3rd Quarter4th Quarter 2 answers
-
Suppose a population of bacteria doubles every hour, but that 1.0 x 106 individuals are removed before reproduction to be converted into valuable biological by-products. Suppose the population begins...
-
Write the equilibrium-constant expression for the equilibrium The table that follows shows the relative mole percentages of CO2(g) and CO(g) at a total pressure of 1 atm for several temperatures....
-
Calculate the percent ionization of propionic acid (C2H5COOH) in solutions of each of the following concentrations (Ka is given in Appendix D): (a) 0.250 M, (b) 0.0800 M, (c) 0.0200 M.
-
Calculate the pH at the equivalence point in titrating 0.100 M solutions of each of the following with 0.080 MNaOH: (a) hydrobromic acid (HBr), (b) chlorous acid (HClO2), (c) benzoic acid (C6H5COOH).
-
Visit SunGards web site, http://www.sungard.com, and research its recovery services offered for the following classes: High Availability, System Recovery, and End-User Recover. Write a report of your...
-
Micro Systems, a developer of database software packages, is a publicly held company and listed with the SEC. The company has no internal audit function. In complying with SOX, Micro Systems has...
-
Leigh Industries has an internal audit department consisting of a director and four staff auditors. The director of internal audit, Diane Bauer, reports to the corporate controller, who receives...

Study smarter with the SolutionInn App


