The malonic ester synthesis is a method for preparing carboxylic acids from alkyl halides. For each...
Fantastic news! We've Found the answer you've been seeking!
Question:
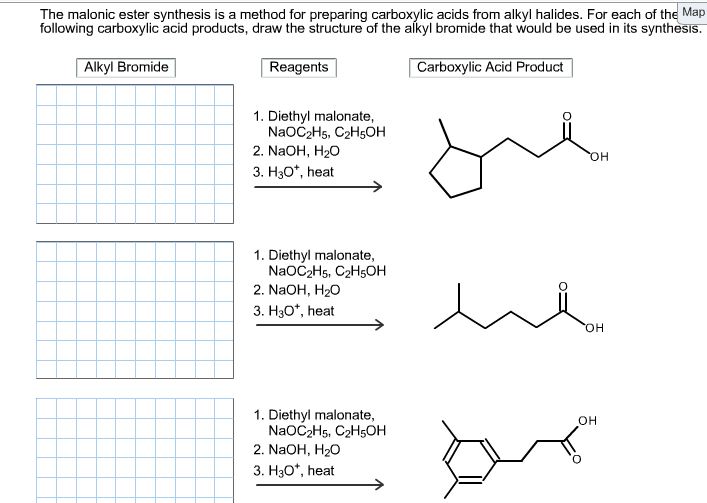
Transcribed Image Text:
The malonic ester synthesis is a method for preparing carboxylic acids from alkyl halides. For each of the Map following carboxylic acid products, draw the structure of the alkyl bromide that would be used in its synthesis. Reagents Carboxylic Acid Product Alkyl Bromide 1. Diethyl malonate, NaOC₂H5, C₂H5OH 2. NaOH, H₂O 3. H3O*, heat 1. Diethyl malonate, NaOC₂H5, C₂H5OH 2. NaOH, H₂O 3. H3O*, heat 1. Diethyl malonate, NaOC2H5, C2H5OH 2. NaOH, H₂O 3. H3O*, heat OH OH OH The malonic ester synthesis is a method for preparing carboxylic acids from alkyl halides. For each of the Map following carboxylic acid products, draw the structure of the alkyl bromide that would be used in its synthesis. Reagents Carboxylic Acid Product Alkyl Bromide 1. Diethyl malonate, NaOC₂H5, C₂H5OH 2. NaOH, H₂O 3. H3O*, heat 1. Diethyl malonate, NaOC₂H5, C₂H5OH 2. NaOH, H₂O 3. H3O*, heat 1. Diethyl malonate, NaOC2H5, C2H5OH 2. NaOH, H₂O 3. H3O*, heat OH OH OH
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Show how you would use the malonic ester synthesis to make the following compounds. (a) (b) (c) CH,CH C-OH
-
The malonic ester synthesis cannot be used to make 2,2-dimethylhexanoic acid. Explain why not.
-
When a malonic ester synthesis is performed using excess base and 1,4-dibromobutane as the alkyl halide, an intramolecular reaction occurs, and the product contains a ring. Draw the product of this...
-
1) Write a generic function to integrate y(x)dx with the following format function I integrator (x, y, method) where x is a vector and y is a matrix whose columns y(:,j) are vectors of the same...
-
Williams Optical Inc. is considering a new just-in-time product cell. The present manufacturing approach produces a product in four separate steps. The production batch sizes are 40 units. The...
-
Northern Airways is about to introduce a daily round-trip flight from New York to Los Angeles and is determining how to price its round-trip tickets. The market research group at Northern Airways...
-
Respondents Varsity Brands, Inc., Varsity Spirit Corporation, and Varsity Spirit Fashions & Supplies, Inc., design, make, and sell cheerleading uniforms. Respondents have obtained or acquired more...
-
Dunbar Distribution markets CDs of numerous performing artists. At the beginning of March, Dunbar had in beginning inventory 2,500 CDs with a unit cost of $7. During March, Dunbar made the following...
-
Date Account Title Debit Credit Sept 1 Cash 10,000 Common Stock 10,000 Sept 2 Equipment 5,000 Cash 5,000 Sept 3 Rent Expense 400 Cash 400 Sept 4 Supplies 500 Cash 500 Sept 5 Advertising Expense 250...
-
The plot below shows the output response of a certain second-order process to a step change in the input. The input is initially at zero and then suddenly decreases at t = 2. The magnitude of the...
-
Caractersticas o elementos relevantes que contemplan la diversificacin de inversiones en el mercado de divisas.
-
Prepare a detailed media on backgrounder Apple Inc. It should be written in narrative format covering the history of the company. It should also list the company's maim consumer technology...
-
Explain how the doctrine of stare decisis not only produces stability and predictability in the common law, but also enables the common law to evolve to meet changing social conditions.
-
If the cost of good sold is 9 6 , 0 0 0 beginning merchandise inventory 8 0 0 0 and merchandise purchase are 1 1 0 , 0 0 0 then What is the ending merchandise inventory? Show your Calculations.
-
A company has the following : Sales ( 6 0 0 units ) $ 4 8 0 , 0 0 0 Variable Expenses $ 2 4 0 , 0 0 0 Fixed Expenses $ 2 0 0 , 0 0 0 What is the net operating income ( profit ) ?
-
Provide a detailed description of the IPhone 13. Information provided MUST include, but is not limited to: history of the product, features of the product, substitutes for the product, complementary...
-
Joe wants to start a savings plan to pay for his children's anticipated college educations. Based on current forecasts, Joe's expenditures on his elder daughter's college education will be $42000 per...
-
The following table shows the rates of total return in successive years from 2004 to 2008 for the Sprott Canadian Equity Fund and for the benchmark Toronto Stock Exchange S&P/TSX Composite Index. By...
-
Indicate the peptides that would result from cleavage by the indicated reagent: a. His-Lys-Leu-Val-Glu-Pro-Arg-Ala-Gly-Ala by trypsin b. Leu-Gly-Ser-Met-Phe-Pro-Tyr-Gly-Val by chymotrypsin c....
-
Rank the following groups of compounds in order of decreasing solubility in water: a. CH3CH3CH3OH CH3CH3CH3CH3Cl CH3CH3CH3CH3OH HOCH3CH3CH3OH b. CH NH2 OH
-
Using curved arrows, show the principal fragments that would be observed in the mass spectrum of each of the following compounds: a. b. c. d. e. f. CH CH CH CH CH OH CH CCH CH2CH2CH3 CH3CH...
-
Some stream data have been collected from a process plant, and these are listed in Table P19.2. Assuming the minimum temperature difference between streams, \(\Delta T_{\min }=10^{\circ} \mathrm{C}\)...
-
Figure P19.3 shows a network design using steam, cooling water and some heat recovery. (a) Does this design achieve the minimum energy target for \(\Delta T_{\min }=20^{\circ} \mathrm{C}\) ? (b) If...
-
Figure P19.4 shows two hot streams and two cold streams for heat integration (subject to \(\Delta T_{\min }=20^{\circ} \mathrm{C}\) ). (i) What are the energy targets? (ii) Show a network design...

Study smarter with the SolutionInn App



