The number of protons, neutrons and electrons for strontium ion: O Protons-38, neutrons 48, electrons -...
Fantastic news! We've Found the answer you've been seeking!
Question:
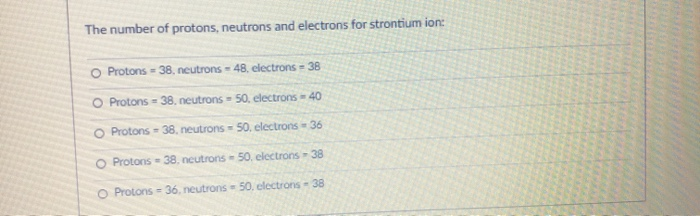


Transcribed Image Text:
The number of protons, neutrons and electrons for strontium ion: O Protons-38, neutrons 48, electrons - 38 O Protons-38, neutrons 50, electrons - 40 O Protons-38, neutrons 50, electrons = 36 O Protons-38, neutrons 50. electrons - 38 O Protons - 36, neutrons 50. electrons - 38 Which of the following statements are correct about modern atomic theory? O Matter is made of small particles called ions. O All atoms of the same element have the same mass. O Different forms of atoms are elements. All atoms of the same element have the same charge. O Atoms cannot be subdivided into smaller particles. Which of the following is not evidence for modern atomic theory? O Dalton's gas ratio analysis O Radioactivity O Atomic force microscopy O Democritus philosophy O Brownian motion The number of protons, neutrons and electrons for strontium ion: O Protons-38, neutrons 48, electrons - 38 O Protons-38, neutrons 50, electrons - 40 O Protons-38, neutrons 50, electrons = 36 O Protons-38, neutrons 50. electrons - 38 O Protons - 36, neutrons 50. electrons - 38 Which of the following statements are correct about modern atomic theory? O Matter is made of small particles called ions. O All atoms of the same element have the same mass. O Different forms of atoms are elements. All atoms of the same element have the same charge. O Atoms cannot be subdivided into smaller particles. Which of the following is not evidence for modern atomic theory? O Dalton's gas ratio analysis O Radioactivity O Atomic force microscopy O Democritus philosophy O Brownian motion
Expert Answer:
Answer rating: 100% (QA)
Protons38 neutrons 48 electrons38 This is because strontium is a chemical element with atomic number ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Which of the following statements are true? 1. Accounting transactions are stored in the operational database. 2. A database administrator is responsible for designing the database. 3. The result of...
-
Which of the following statements are true regarding dividends? _____ 1. Cash and stock dividends reduce retained earnings. _____ 2. Dividends payable is recorded at the time a cash dividend is...
-
Which of the following statements are false? (a) Dipole-dipole interactions between molecules are greatest if the molecules possess only temporary dipole moments. (b) All compounds containing...
-
Midwest Inc. is a medium-size company that has been in business for 20 years. The industry has become very competitive in the last few years, and Midwest has decided that it must grow if it is going...
-
The chapter describes how the dividends valuation approach measures value-relevant dividends to encompass various transactions between the firm and the common shareholders. What transactions should...
-
Born-again Christian groups have been instrumental in organizing boycotts of products advertised on shows they find objectionable, especially those that they feel undermine family values. Do...
-
Cloud computing has just begun to take off in the business world. The biggest player in the cloud computing marketplace is one you might not expect: Amazon. Under its Web Services division (AWS),...
-
1. Discuss the pros and cons of using average demand to assess capacity requirements. Is this a reasonable approach for the ER? 2. Make a recommendation for Jenn as to what she should do and the...
-
Howell Petroleum, Incorporated, is trying to evaluate a generation project with the following cash flows: Year 1 2 Cash Flow -$52,000,000 74,000,000 -12,000,000 a-1. What is the NPV for the project...
-
K Dhoni is a business consultant. The following trial balance is provided at 30 September 201: The following additional information is supplied: 1. At 30 September 201 rent received in advance...
-
Consider a triangle ABC like the one below. Suppose that b=67, a=33, and B = 109. (The figure is not drawn to scale.) Solve the triangle. Carry your intermediate computations to at least four decimal...
-
What efforts have South American countries made to regionally integrate their economies?
-
What is the Caribbean Basin Initiative? What is its goal?
-
Discuss the primary modes of entering a foreign market. What are the advantages and disadvantages of each of these modes?
-
What strategies can North American and Asian firms adopt to ensure access to the enormous EU market?
-
What factors should be considered in selecting a strategic alliance partner?
-
the TED talk "The surprising connection between brain injuries and crime" byKim Gorgens 1) What does Dr. Gorgens mean by "serving a life sentence 30 days at a time"? (Please describe sequence of...
-
The May 2014 revenue and cost information for Houston Outfitters, Inc. follow: Sales Revenue (at standard).............. $ 540,000 Cost of Goods Sold (at standard) ..........341,000 Direct Materials...
-
(a) Calculate the number of molecules in a deep breath of air whose volume is 2.25 L at body temperature, 37oC, and a pressure of 735 torr. (b) The adult blue whale has a lung capacity of 5.0 x 103...
-
At an underwater depth of 250 ft, the pressure is 8.38 atm. What should the mole percent of oxygen be in the diving gas for the partial pressure of oxygen in the mixture to be 0.21 atm, the same as...
-
Serotonin is a compound that conducts nerve impulses in the brain. It contains 68.2 mass percent C, 6.86 mass percent H, 15.9 mass percent N, and 9.08 mass percent O. Its molar mass is 176 g/mol....
-
You want to lease a set of golf clubs from Pings Ltd. for \($4,000.\) The lease contract is in the form of 24 months of equal payments at a 12% annual percentage rate (APR). Suppose payments are due...
-
Ernie Els wants to save money to meet two objectives. First, he would like to be able to retire 30 years from now with a retirement income of \($300,000\) per year for 20 years beginning at the end...
-
A 10-year annuity pays \($900\) per year, with payments made at the end of each year. The first \($900\) will be paid 5 years from now. If the APR is 8% and interest is compounded quarterly, what is...

Study smarter with the SolutionInn App


