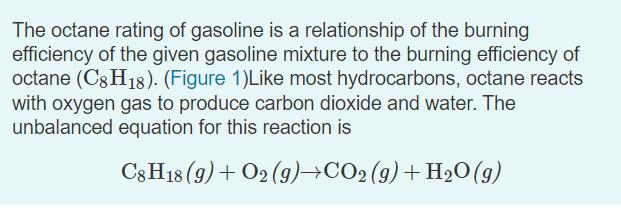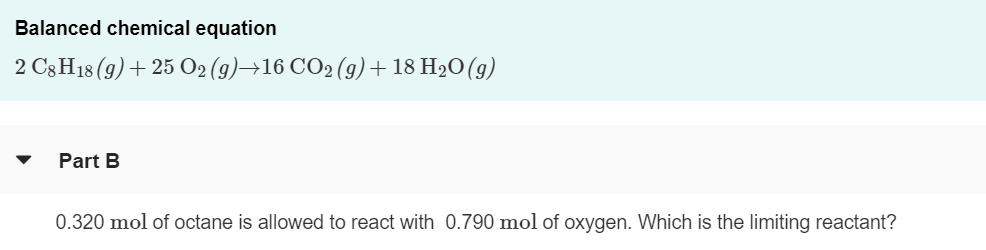The octane rating of gasoline is a relationship of the burning efficiency of the given gasoline...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
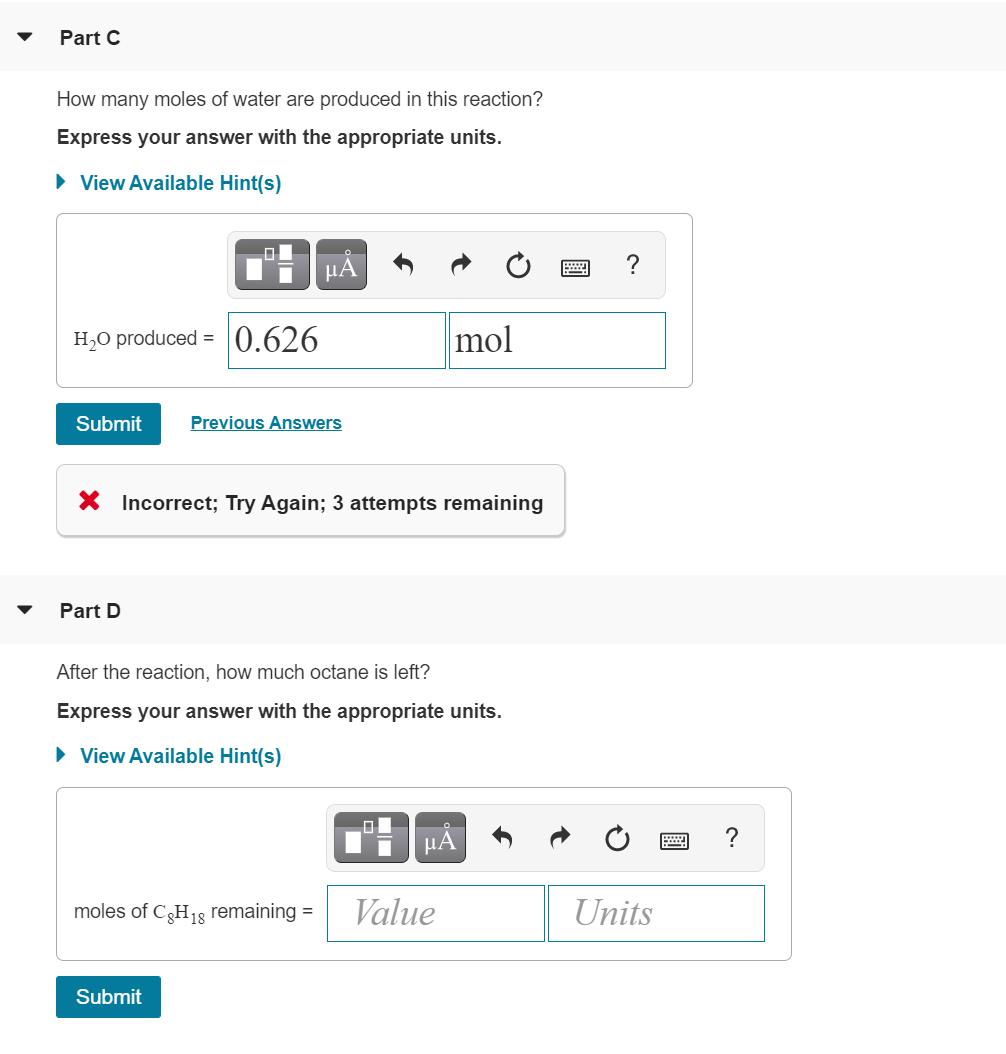
The octane rating of gasoline is a relationship of the burning efficiency of the given gasoline mixture to the burning efficiency of octane (C8 H18). (Figure 1)Like most hydrocarbons, octane reacts with oxygen gas to produce carbon dioxide and water. The unbalanced equation for this reaction is C3 H18 (9) + O2 (9)+CO2 (9)+ H2O(g) Balanced chemical equation 2 C3H18 (9)+ 25 O2 (9)→16 CO2 (9) + 18 H2O(g) Part B 0.320 mol of octane is allowed to react with 0.790 mol of oxygen. Which is the limiting reactant? Part C How many moles of water are produced in this reaction? Express your answer with the appropriate units. • View Available Hint(s) HẢ ? H,O produced = 0.626 mol Submit Previous Answers X Incorrect; Try Again; 3 attempts remaining Part D After the reaction, how much octane is left? Express your answer with the appropriate units. > View Available Hint(s) µA moles of CH18 remaining = Value Units Submit The octane rating of gasoline is a relationship of the burning efficiency of the given gasoline mixture to the burning efficiency of octane (C8 H18). (Figure 1)Like most hydrocarbons, octane reacts with oxygen gas to produce carbon dioxide and water. The unbalanced equation for this reaction is C3 H18 (9) + O2 (9)+CO2 (9)+ H2O(g) Balanced chemical equation 2 C3H18 (9)+ 25 O2 (9)→16 CO2 (9) + 18 H2O(g) Part B 0.320 mol of octane is allowed to react with 0.790 mol of oxygen. Which is the limiting reactant? Part C How many moles of water are produced in this reaction? Express your answer with the appropriate units. • View Available Hint(s) HẢ ? H,O produced = 0.626 mol Submit Previous Answers X Incorrect; Try Again; 3 attempts remaining Part D After the reaction, how much octane is left? Express your answer with the appropriate units. > View Available Hint(s) µA moles of CH18 remaining = Value Units Submit The octane rating of gasoline is a relationship of the burning efficiency of the given gasoline mixture to the burning efficiency of octane (C8 H18). (Figure 1)Like most hydrocarbons, octane reacts with oxygen gas to produce carbon dioxide and water. The unbalanced equation for this reaction is C3 H18 (9) + O2 (9)+CO2 (9)+ H2O(g) Balanced chemical equation 2 C3H18 (9)+ 25 O2 (9)→16 CO2 (9) + 18 H2O(g) Part B 0.320 mol of octane is allowed to react with 0.790 mol of oxygen. Which is the limiting reactant? Part C How many moles of water are produced in this reaction? Express your answer with the appropriate units. • View Available Hint(s) HẢ ? H,O produced = 0.626 mol Submit Previous Answers X Incorrect; Try Again; 3 attempts remaining Part D After the reaction, how much octane is left? Express your answer with the appropriate units. > View Available Hint(s) µA moles of CH18 remaining = Value Units Submit The octane rating of gasoline is a relationship of the burning efficiency of the given gasoline mixture to the burning efficiency of octane (C8 H18). (Figure 1)Like most hydrocarbons, octane reacts with oxygen gas to produce carbon dioxide and water. The unbalanced equation for this reaction is C3 H18 (9) + O2 (9)+CO2 (9)+ H2O(g) Balanced chemical equation 2 C3H18 (9)+ 25 O2 (9)→16 CO2 (9) + 18 H2O(g) Part B 0.320 mol of octane is allowed to react with 0.790 mol of oxygen. Which is the limiting reactant? Part C How many moles of water are produced in this reaction? Express your answer with the appropriate units. • View Available Hint(s) HẢ ? H,O produced = 0.626 mol Submit Previous Answers X Incorrect; Try Again; 3 attempts remaining Part D After the reaction, how much octane is left? Express your answer with the appropriate units. > View Available Hint(s) µA moles of CH18 remaining = Value Units Submit
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
In a refinery, the octane rating of gasoline produced is measured by taking one observation from each batch. Twenty observations follow. a. Construct Three Sigma control charts for the individual...
-
How many moles of BH3 are needed to react with 2 moles of 1-pentene?
-
How many moles of water are there in 1.000 L? How many molecules?
-
Perpetual inventory using FIFO Beginning inventory, purchases, and sales for Item Zeta9 are as follows: Oct. 1 Inventory 37 units @ $19 Oct. 7 Sale Oct. 15 Purchase Oct. 24 Sale Assuming a perpetual...
-
Consider the measurable space ((i, Ai), i = 1, 2, and let C be the class of all countable sums of rectangles (unions of pair-wise disjoint rectangles) in the product space (1 (2. Then by an example,...
-
What is GSS?
-
Electric motors generate heat in their rotor. What is the average time for a loaded motor to reach stable temperature?
-
David Tennant has prepared the following list of statements about partnerships. 1. A partnership is an association of three or more persons to carry on as co-owners of a business for profit. 2. The...
-
Answer the following questions about exchanging information with families about their children. For each procedure listed below, explain how it helps educators and families exchange information about...
-
In 1879, A.A. Michelson measured the velocity of light in air using a modification of a method proposed by the French physicist Foucault. Twenty of these measurements are in table 6E.27 (the value...
-
What is the purpose of using a UDP ping scan in network security? a. The purpose of using a UDP ping scan in network security is to block unauthorized access to a network. b. The purpose of using a...
-
Indirect cost rate is the ratio of what to what for a given time period? A. Indirect expense to indirect cost. B. An appropriate base to indirect cost. C. Indirect expense to an appropriate base. D....
-
5. (a) Say you have a HashMap of size 1000 and one search operation takes approxi- mately 1ms. How long will one search take (approximately, on average) on a HashMap of size 2000? (b) Same question,...
-
C wants to impress D with a gift that will induce D to marry C. C purchases jewelry from E that, much to C's surprise, is ersatz. Discuss C's rights against E when D laughs in C's face. Would C's...
-
Explain the impact(s) of the US withdrawal from the TPP on the relationship between NAFTA/USMCA members. For instance, if US administration is currently no more a member of the TPP, have they...
-
A key element of torts analysis is the duty of care that a business owes to its customers and other stakeholders. If such a duty exists, breach of that duty can lead to liability for harm from the...
-
X Corporation is a company involved in manufacturing mining equipment. At the beginning of the year, the board of directors of the said company has decided to enter into a business combination with Y...
-
Solve for the equilibria of the following discrete-time dynamical systems Pr pt+1 = Pr+2.0(I-Pr)
-
Which of the following reactions lies to the right, favoring the formation of products, and which lies to the left, favoring formation of reactants? (a) (b) 2 NO(g) + O2(g) =2 NO,(g); K, = 5.0 X 1012
-
Brass is a substitutional alloy consisting of a solution of copper and zinc. A particular sample of red brass consisting of 80.0% Cu and 20.0% Zn by mass has a density of 8750 kg/m3. (a) What is the...
-
Explain how the electron-sea model accounts for the high electrical and thermal conductivity of metals?
-
Why are none of the bulbs in Figure 31. 24 lit? Data from Figure 31. 24 Figure 31.24 (1) (iii) NNN
-
In Figure 31. 25, identify the energy conversions that occur between points \(A\) and \(B, B\) and \(\mathrm{C}, \mathrm{C}\) and \(\mathrm{D}\), and \(\mathrm{D}\) and \(\mathrm{A}\). Data from...
-
In Figure 31. 26, bulb B is brighter than bulb C, which in turn is brighter than bulb A. Rank, largest first, \((a)\) the magnitudes of the potential differences across the bulbs, \((b)\) the...

Study smarter with the SolutionInn App