The photoelectric threshold wavelength of silver is 3250 x 10 m. The velocity of the electron...
Fantastic news! We've Found the answer you've been seeking!
Question:
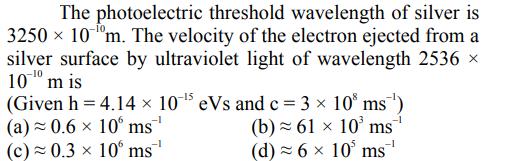
Transcribed Image Text:
The photoelectric threshold wavelength of silver is 3250 x 10 m. The velocity of the electron ejected from a silver surface by ultraviolet light of wavelength 2536 10 m is (Given h = 4.14 10-5 eVs and c = 3 10 ms ) (a) 0.6 x 10 ms (b) 61 x 10 ms (c) 0.3 x 10 ms -1 (d) = 6 x 10 ms The photoelectric threshold wavelength of silver is 3250 x 10 m. The velocity of the electron ejected from a silver surface by ultraviolet light of wavelength 2536 10 m is (Given h = 4.14 10-5 eVs and c = 3 10 ms ) (a) 0.6 x 10 ms (b) 61 x 10 ms (c) 0.3 x 10 ms -1 (d) = 6 x 10 ms
Expert Answer:
Related Book For 

Probability & Statistics For Engineers & Scientists
ISBN: 9780130415295
7th Edition
Authors: Ronald E. Walpole, Raymond H. Myers, Sharon L. Myers, Keying
Posted Date:
Students also viewed these physics questions
-
The photoelectric threshold wavelength of a tungsten surface is 272 run. Calculate the maximum kinetic energy of the electrons ejected from this tungsten surface by ultraviolet radiation of frequency...
-
Ultraviolet light of wavelength 220 nm illuminates a tungsten surface and electrons are ejected. A stopping potential of 1.1 V is able to just prevent any of the ejected electrons from reaching the...
-
The photoelectric effect threshold wavelength for a certain metal is 400 nm. Calculate the maximum speed of photoelectrons if we use light having a wavelength of (a) 300 nm, (b) 400 nm, and (c) 500...
-
Many auditors consider the substantiation of the figure for inventory to be a more difficult and challenging task than the verification of most other items on the balance sheet. List several specific...
-
Accounting for forward foreign exchange contract as a cash flow hedge. On October 1, 2008, Riddle Corporation purchases equipment from a supplier in France on account at a purchase price of 40,000...
-
If the input is -5, what is the output? If x less than 0 Put "Low " to output Else if x greater than 0 Put "OK " to output Else if x less than -3 Put "Very low " to output a) Low b) Low Very low c)...
-
Figure 10-22 depicts the activities performed in the revenue cycle by the Newton Hardware Company. Required 1 Identify the weaknesses in Newton Hardwares revenue cycle. Explain the resulting threat...
-
Blair Gasses and Chemicals is a supplier of highly purified gases to semiconductor manufacturers. A large chip producer has asked Blair to build a new gas production facility close to an existing...
-
Define business ethics and corporate social responsibility ( CSR ) . How can ethical decision - making and CSR initiatives benefit an organization's reputation and long - term success?
-
Refer the following table. Focus Metals Inc. Comparative Balance Sheet Information: November 30 (millions of $) 2020 Cash 16 86 Accounts receivable (net). 386 232 60 53 Inventory Plant and equipment...
-
! Required information The graph shows vx vs. t for an object moving along the x-axis. vx (m/s) 40 20 0 2 4 6 8 10 12 141(s) What is ax at t = 1.00 s? m/s
-
How would advocates of the Taylor Rule or Monetarism probably feel about the Fed's policy moves during the terms of Fed Chairs Bernanke, Yellen, and Powell? What is currency manipulation? Why would a...
-
Items from the 20x1 income statement, statement of retained earnings, and balance sheet of ABC, Inc., are listed below in alphabetical order. Solve for the missing amounts. (Loss amounts should be...
-
Terry is considering attending the Culinary Institute ofAmerica in New York City. It is a four-year program with tuitioncosts of $29,380 per year. He currently makes $55,000 per year as asous chef,...
-
Contribute to safe workplace practices in the workplace by raising health and safety issues with designated persons in line with workplace procedures. What does an individual do to contribute?
-
How much more energy does a 73 nm photon have than a 4070 nm photon? Does the 73 nm photon move at a greater speed than the 4070 nm photon? Which photon has a higher frequency? Part 1 of 4 The energy...
-
two charged point particles are placed in the x - y plane as shown. the charges and co - ordinates are: q 1 = + 3 . 0 C , x 1 = - 2 cm , y 1 = 0 and q 2 = - 5 C , x 2 = + 2 cm , y 2 = 4 cm . Find the...
-
What is the order p of a B + -tree? Describe the structure of both internal and leaf nodes of a B + -tree.
-
How fast must an electron move to have a kinetic energy equal to the photon energy of sodium light at wavelength 590 nm?
-
The existence of the atomic nucleus was discovered in 1911 by Ernest Rutherford, who properly interpreted some experiments in which a beam of alpha particles was scattered from a metal foil of atoms...
-
A taxi company manager is trying to decide whether the use of radial tires instead of regular belted tires improves fuel economy. Twelve cars were equipped with radial tires and driven over a...
-
Estimate the ending inventory by the gross profit method, given the following data: beginning inventory, \(\$ 40,000\); net purchases, \(\$ 100,000\); net sales, \(\$ 112,000\); average gross profit...
-
Use your operating systems built-in capability to encrypt a file. Store the enciy'pted file in a folder that can be shared with other users on that computer. Then create a new user account. Assign it...
-
Why do companies using LIFO in a perpetual inventory system often restate their ending inventory at the per-unit costs that result from applying periodic LIFO costing procedures?

Study smarter with the SolutionInn App



