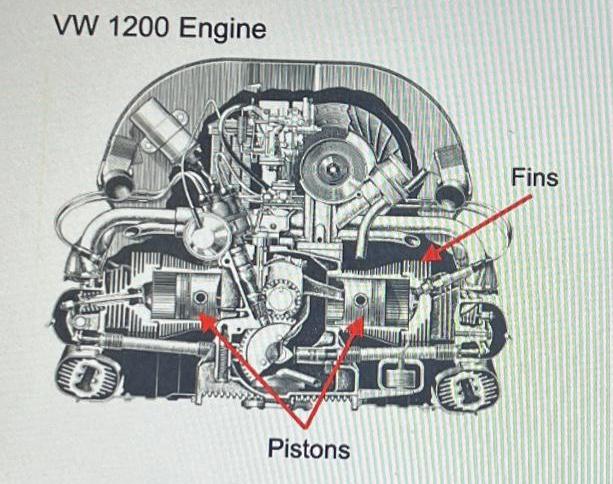
The picture above shows the VW 1200 Engine, which was used in the VW Beetle The...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
The picture above shows the VW 1200 Engine, which was used in the VW Beetle The car had the tendency to overheat on hot days at rest stops. Explain the role of the fins on the motor and explain, what you could do to improve the cooling of the engine. VW 1200 Engine Pistons Fins The picture above shows the VW 1200 Engine, which was used in the VW Beetle The car had the tendency to overheat on hot days at rest stops. Explain the role of the fins on the motor and explain, what you could do to improve the cooling of the engine. VW 1200 Engine Pistons Fins
Expert Answer:
Answer rating: 100% (QA)
The fins on the VW 1200 engine commonly used in the VW Beetle serve a critical role in regulating the engines temperature These fins are part of the e... View the full answer

Related Book For 

Management A Practical Introduction
ISBN: 978-0078112713
5th edition
Authors: Angelo Kinicki, Brian Williams
Posted Date:
Students also viewed these accounting questions
-
Analysis on Apple Computer, and additional research, describe what SCM strategies Apple applies, including the use of Apple Stores, and if they can stand the test of time/competition in SCM?
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Oriole Company uses a job order cost system and applies overhead to production on the basis of direct labor costs. On January 1 , 2 0 2 5 , Job 5 0 was the only job in process. The costs incurred...
-
In Exercises 3344, use the graph of y = f(x) to graph each function g. g(x) = f(x) - 2 -4,0) -5-4-3 4-33 y = f(x) y (0,0) 2- 3.4 45 -2) (4-2) X
-
A hydrogen atom is in the initial state ni = n, where n > 1. (a) Find the frequency of the photon that is emitted when the electron jumps to state nf = n - 1. (b) Find the frequency of the electron's...
-
The following data on y = glucose concentration (g/L) and x = fermentation time (days) for a particular blend of malt liquor was read from a scatterplot in the article "Improving Fermentation...
-
Automakers provide an interesting study in cost management strategies. General Motors often provides a contrast to other U.S. manufacturers in that approximately 46,000 of its workers are...
-
Schylar Pharmaceuticals, Inc., plans to sell 130,000 units of antibiotic at an average price of $22 each in the coming year. Total variable costs equal $1,086,800. Total fixed costs equal $8,000,000....
-
Consider the following list of internal controls. For each control classify it either as a preventive control or a detective control. Also classify each control as either a manual control, an IT...
-
Fawcett Institute provides one-on-one training to individuals who pay tuition directly to the business and also offers extension training to groups in off-site locations. Fawcett prepares adjusting...
-
A stock had the following annual returns: - 1 1 . 0 6 % , 8 . 9 5 % , 1 2 . 9 8 % , and 1 3 . 9 5 % . Compute the following for the stock: Expected Return ( Please write your answer as a percentage (...
-
6. If an airplane does a vertical loop in the air, with a circular arc of radius 273 meters, then what speed does the plane have to go for the pilot to experience (a) versus (b) as stated below. [...
-
You are making an audit of the shareholder's equity accounts of Clarence Corporation for the year ended December 31, 2017 The statement of financial position at December 31, 2016 showed the following...
-
Calculate the magnitude (signs don't matter) of the centripetal acceleration in an electron if it is moving around a nucleus with a speed of 2.18 x 10 m/s. The diameter of the electron's orbit is...
-
If you had to speak in front of the Detroit City Council about this issue, who would you be supporting - the Hagermans or Judy Kelley? Explain your answer. People Are Making Big Money Kicking Detroit...
-
Why aren't all sports balls made of the same material? Choose a sport that uses a ball. Explain how you think that ball is designed for that sport. Share what you have learned from your own...
-
Review the annual reports of PepsiCo and Amazon, two of the largest publicly traded firms in America. Compute and compare the accounts receivable turnover ratios and the average collection period in...
-
6 (a) Briefly develop a mathematical model of the behaviour of a copper-twisted pair cable (b) Derive the magnetic energy from: w given that: K + w, where the - - k symbols have their usual meaning...
-
I. Organizations should stay out of our personal lives. If we want to overeat or smoke, that is our choice. Organizations should not be allowed to monitor weight and smoking behavior. 2....
-
1. How many of the five sources of power is Mark Zuckerberg using? Explain. 2. Which of the eight influence tactics is Zuckerberg using? 3. Which different leadership traits and styles does...
-
Your brother-in-law just submitted a purchase request for materials valued at $10,000 for side jobs. What should you do? 1. Approve the purchase. It's not your job to police your brother-in-law, and...
-
On January 1, 2011, BGA Company had a balance of \(\$ 500,000\) in its Bonds Payable account. During 2011, BGA issued bonds with a \(\$ 150,000\) face value. There was no premium or discount...
-
The following accounts and corresponding balances were drawn from Winston Company's 2012 and 2011 year-end balance sheets. Other information drawn from the accounting records: 1. Winston incurred a...
-
Top Brands, Inc. (TBI), presents its statement of cash flows using the indirect method. The following accounts and corresponding balances were drawn from TBI's 2012 and 2011 year-end balance sheets....

Study smarter with the SolutionInn App


