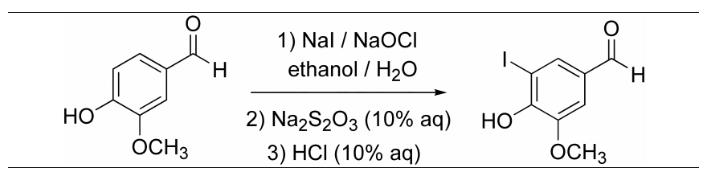
The pKa of the starting material (Ar-OH) is ~9, which is much more acidic than a normal
Fantastic news! We've Found the answer you've been seeking!
Question:
The pKa of the starting material (Ar-OH) is ~9, which is much more acidic than a normal alkyl alcohol (pKa ~15)? The reason is resonance stabilization of the conjugate base. Please draw the deprotonation of the vanillin, and no less than 5 resonance structures of the conjugate base to explain the enhanced acidity of the phenol. (There is also a 6th one, can you find it?)
Related Book For 

Posted Date:




