The product of atomic mass and metal specific heat is about 6.4. This information was provided...
Fantastic news! We've Found the answer you've been seeking!
Question:
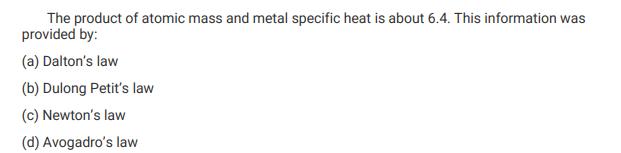
Transcribed Image Text:
The product of atomic mass and metal specific heat is about 6.4. This information was provided by: (a) Dalton's law (b) Dulong Petit's law (c) Newton's law (d) Avogadro's law The product of atomic mass and metal specific heat is about 6.4. This information was provided by: (a) Dalton's law (b) Dulong Petit's law (c) Newton's law (d) Avogadro's law
Expert Answer:
Related Book For 

Accounting concepts and applications
ISBN: 978-0538745482
11th Edition
Authors: Albrecht Stice, Stice Swain
Posted Date:
Students also viewed these chemistry questions
-
The following information was provided by Hitchens widget store for July 31, 202X. They make two products: R2D2 & C3P0. Both items use 2 of the 3 listed materials: Nada, Yada, and Bozo. Presented...
-
The following information was provided by Sibongile (Pty) Ltd for Product Alpha. This information relates to the second quarter, which has just ended. Budgeted production was 90 000 units and 100 000...
-
The following balance sheet information was provided by O'Connor Company: Assets Year 2 Year 1 Cash $2,000 $1,000 Accounts receivable $7,000 $ 5,000 Inventory $20,000 $21,000 Assuming that net credit...
-
Use Gordon formula to calculate the price of the stock. ITC company expected to pay dividend of $4 and growth rate of 3% per year is constant forever. Required return is 8%.
-
Why is practical capacity recommended in calculating capacity cost rates?
-
A man drags a 72-kg crate across the floor at a constant velocity by pulling on a strap attached to the bottom of the crate. The crate is tilted 25 above the horizontal, and the strap is inclined 61...
-
Calculate \(2+3^{2} \times 4\).
-
Mega Electronix sells television sets and DVD players. The business is divided into two divisions along product lines. CVP income statements for a recent quarter's activity are presented below....
-
Anwer 5 MCQ with simple explanations:. 6. Identify the true statement about currency futures contracts a. b. c. Futures contracts are not traded in organized markets. (1. E}. The design of futures...
-
Comprehensive Problem 1 - Part 1: Taxpayer information, Form 1040, Schedules 1, 2, 3 and 4, Schedule A, and Schedule B. Noah and Joan Arc's Tax Return Note: This problem is divided into three parts....
-
How do I build a supply chain plan/diagram for a new business by analyzing factors that affect sourcing, logistics, metrics, suppliers, and risk? I dont understand the sourcing, logistics and metrics
-
At December 31, Folgeys Coffee Company reports the following results for its calendar year. Cash sales Credit sales $920,000 320,000 Its year-end unadjusted trial balance includes the following...
-
write the output #include using namespace std; int main() { int *a, n: a = new int[10]; for (int i=0; i <10; i++) int *ptr: *(a+1) = i + 2; int *k = 0; int *z: ptr a: k=ptr= ptr=++k; cout
-
On the basis of relationship between the center and the units, the governments may be classified as unitary and federal. Does the Republic of Mauritius operate a federal or unitary system? Justify...
-
17. Given an aluminum wire conductor (power line for example): po=2.63E-8 (Nm) @ To=0C and = 4.4E-3 (deg). What is the percentage increase in resistance between 68F and 120F (A very hot afternoon in...
-
Discuss the role of focalization in shaping narrative perspective and reader interpretation. How do authors like James Joyce or Virginia Woolf use shifting points of view to create a...
-
A data set lists the number of battery charger sales made at a technology store each day for the past few days. For this data set, the minimum is 1 , the first quartile is 11 , the median is 13 , the...
-
Review Exhibit 11.4. Analyze each product on the graph according to the characteristics that influence the rate of adoption. For example, what can you conclude from the data about the relative...
-
Romulus, Inc., issued $500,000 of 10%, five-year bonds at face value on July 1, 2012. Interest on the bonds is payable semiannually on December 31 and June 30. 1. Provide the journal entry to record...
-
Refer to the data in PE 18-7. Using the following additional information, compute the direct labor cost. Direct labor hours per unit . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Which one of the following statements is false? a. Debentures are bonds that have no underlying assets pledged as collateral to guarantee their payment. b. Serial bonds mature in one single sum on a...
-
The following information relates to Heather Limited and its subsidiary, Thistle*Limited. 1. Heather Limited Retained profits as at 31 March 2008 700,000. 80,000 ordinary shares were purchased in...
-
Draw up a consolidated balance sheet as at 31 December 2007 from the following: At the balance sheet date, Child owes Parent 3,200. During the year Parent sold goods which had cost 6,000 to Child for...
-
You are to draw up a consolidated balance sheet as at 31 December 2005 from the following: At the balance sheet date, Son 1 owed Pa and Mum 2,500 and Son 2 1,100, and Pa and Mum owed Son 2 2,100. Pa...

Study smarter with the SolutionInn App



