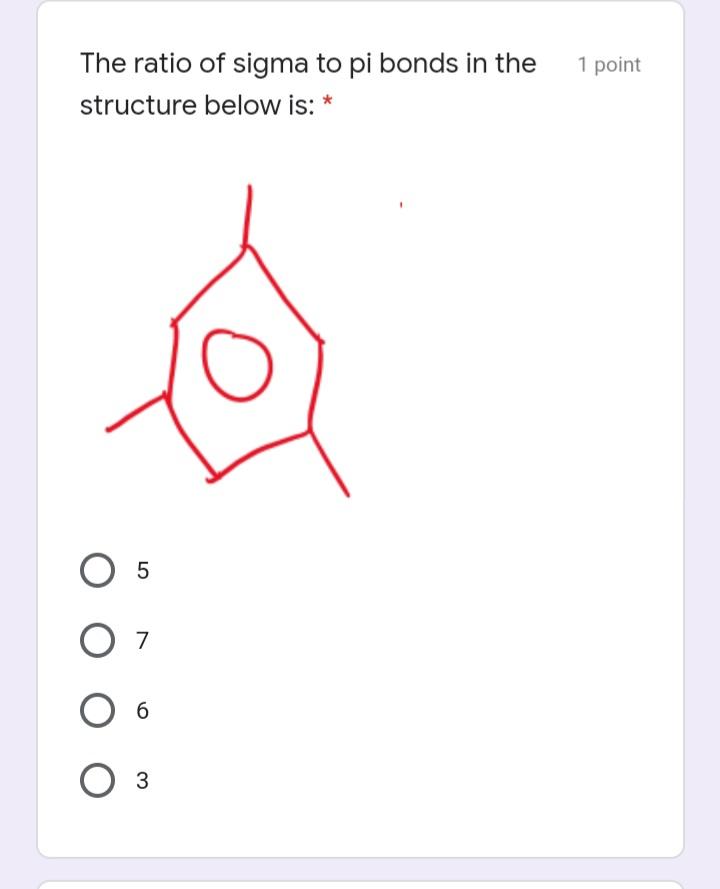
The ratio of sigma to pi bonds in the 1 point structure below is: * O...
Fantastic news! We've Found the answer you've been seeking!
Question:






Transcribed Image Text:
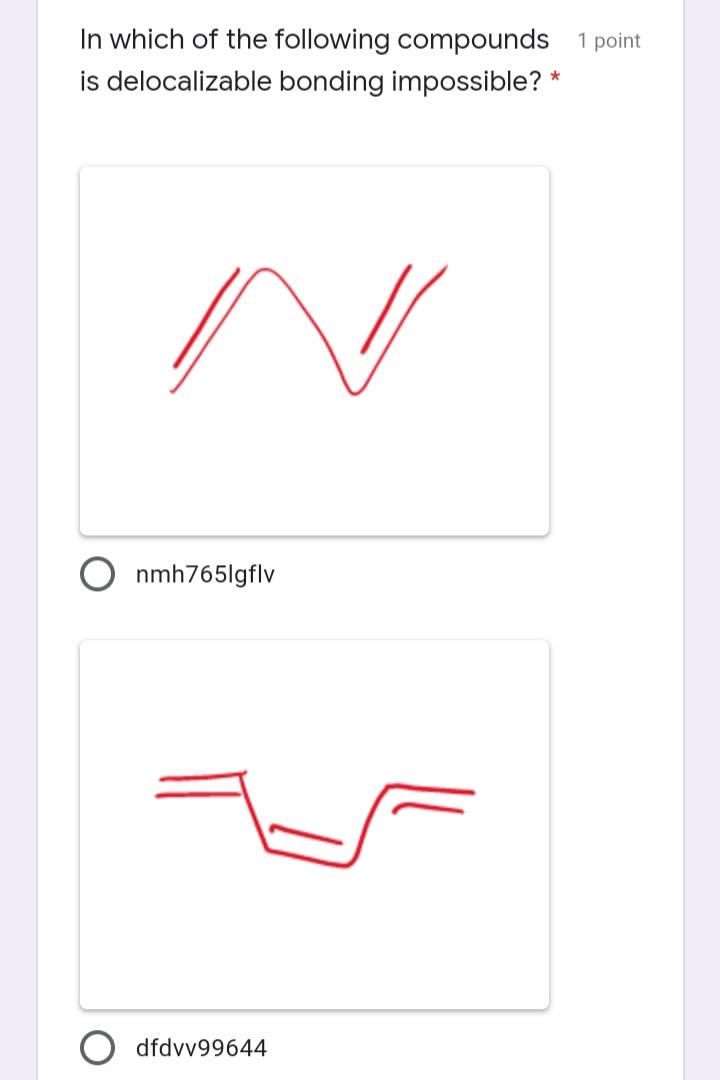
The ratio of sigma to pi bonds in the 1 point structure below is: * O 7 6. 3 In which of the following compounds 1 point is delocalizable bonding impossible? * nmh765lgflv dfdvv99644 O dfbgj89965tgf vcfrerk4353 Where are the two lone pairs of 1 point electrons of the oxygen atom in the molecule given below located? * in two s orbitals in two sp3 orbitals in two sp orbitals in one sp3 orbital O in two sp2 orbitals Which of the following is not a 1 point resonance structure of the molecule below? * NH, E H I. NH2 III. NH2 E H. H. NH2 П. NH2 IV. E E H. H IV II Which of the following is the most 1 point stable? * I. IV. V. П. III. O Iv II O II Which of the following choices 1 point correctly represents the following compound: * (cH3)2 CH CH3 CH CH CHy CH3 CH3 CH3 H. CH,CH3 ffff754redv fefeeet CH3 H CH3 H CH(CH3)2 ffrh98uyg CH3 H CH3 CH5 CH(CH3)2 ffff986rg The ratio of sigma to pi bonds in the 1 point structure below is: * O 7 6. 3 In which of the following compounds 1 point is delocalizable bonding impossible? * nmh765lgflv dfdvv99644 O dfbgj89965tgf vcfrerk4353 Where are the two lone pairs of 1 point electrons of the oxygen atom in the molecule given below located? * in two s orbitals in two sp3 orbitals in two sp orbitals in one sp3 orbital O in two sp2 orbitals Which of the following is not a 1 point resonance structure of the molecule below? * NH, E H I. NH2 III. NH2 E H. H. NH2 П. NH2 IV. E E H. H IV II Which of the following is the most 1 point stable? * I. IV. V. П. III. O Iv II O II Which of the following choices 1 point correctly represents the following compound: * (cH3)2 CH CH3 CH CH CHy CH3 CH3 CH3 H. CH,CH3 ffff754redv fefeeet CH3 H CH3 H CH(CH3)2 ffrh98uyg CH3 H CH3 CH5 CH(CH3)2 ffff986rg
Expert Answer:
Answer rating: 100% (QA)
6 The structure of CHCHCHCHCHCH we see the structure fr... View the full answer

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
What is the shear capacity of the RC beam described below considering the steel reinforcement and using the formula: VRsyAw 2fyd cot 8/s The shear reinforcement in the beam is provided by sets of...
-
In what orbitals are the electrons represented as lone pairs when drawing the structures of quinoline, indole, imidazole, purine, and pyrimidine?
-
The sp2 hybrid atomic orbitals have the following general form: where Ïs, Ïpx, and Ïpy represent orthonormal (normalized and orthogonalized) atomic orbitals. Calculate the values of A...
-
The number x of bicycle helmets people are willing to buy per week from a retail chain at a price of $p is given by x = 1,000 - 60p + 25 20 ¤ p ¤ 100 (see the figure). (A) Find dx/dp....
-
A Western Star over-the-road tractor is purchased for $132,895 and placed in service in July 2010. The owner elects to depreciate this 3-year property using MACRS. Make a depreciation schedule...
-
What makes a posttest-only design "stronger" in terms of internal validity than a design with a pretest? Is it more appropriate to include random assignment for these designs? Why or why not?
-
A balanced orthotropic, or square symmetric lamina, is made up of \(0^{\circ}\) and \(90^{\circ}\) fibers woven into a fabric and bonded together, as shown in Figure 2.10. a. Describe the...
-
Kenoly Corporation owns a patent that has a carrying amount of $300,000. Kenoly expects future net cash flows from this patent to total $210,000 over its remaining life of 10 years. The recoverable...
-
Provide a constructive comparison between the scientific management Human Relations human resource approach as well as the strategic human resource management approach?
-
Complete Form SS-4 for TCLH Industries. The company was formed on December 1, 2019, as a corporation (which files Form 1120S and was incorporated in North Carolina) by Michael Sierra (CEO; SSN 232-...
-
AIM: Write C program to access elements using pointer.
-
TABLE 1-2: Present value of an ordinary annuity of n payments of 1: (P/A, i, n) 1 (1 + i)" i P/A= n 2% 3% 5% 7% 2.5% 10% 12% 14% 15% 5.60143 5.50813 6.47199 6.34939 7.32548 7.17014 8.16224 7.97087...
-
A rocket moves with a velocity: v = At + B where A = 1 5 . 0 m / s ^ 2 , B = 8 . 0 m / s , and t is the time in seconds. At time t = 2 . 0 s , the rocket is located at position x = 5 0 . 0 m . Write...
-
A car moves with velocity: v = At ^ 2 where A = 2 . 0 m / s ^ 3 , and t is the time in seconds. At time t = 3 . 0 s , the car is located at position x = 5 0 . 0 , . What was the car's position at...
-
1. (8 pts.) The following stack stores a stack of integers. Draw the stacks and determine what is the output of the following program? import java.util.*; public class TestStack1 { public static void...
-
A runner in a race moves with velocity: v = At ^ 3 It is known that the runner travels a distance of 1 5 . 0 m during the time interval from 2 . 0 s to 4 . 0 s . What is the value of the constant A ?
-
With the Help of Macaulay's Method Calculating the Deflection at point B and Point C and also Calculate the location of Maximum Deflection and it's value for Given Simply Supported Beam?
-
1. Below is depicted a graph G constructed by joining two opposite vertices of C12. Some authors call this a "theta graph" because it resembles the Greek letter 0. a. What is the total degree of this...
-
Which oxidizing or reducing agent would you use to carry out the following transformations? (a) (b) (c) (d) (e) OCH3 OCH3 OCH3 HHO HHO
-
Give a structure for compound F that is consistent with the 1H NMR spectrum in Fig. 14.28 and IR absorptions at 3020, 2965, 2940, 2870, 1517, 1463, and 818 cm-1.
-
Write structures for the following bicyclic alkanes: (a) Bicyclo [1.1.0] butane (b) Bicyclo [2.1.0] pentane (c) 2-Chlorobicyclo [3.2.0] heptane (d) 7-Methylbicyclo [2.2.1] heptane
-
A thin stainless steel disk of thickness \(b\) and outer radius \(r_{o}\) has been heat treated to a high, uniform initial temperature of \(T_{i}\). The disk is then placed upon a small stand and...
-
A plane wall of a furnace is fabricated from plain carbon steel \(\left(k=60 \mathrm{~W} / \mathrm{m} \cdot \mathrm{K}, ho=7850 \mathrm{~kg} / \mathrm{m}^{3}, c=430 ight.\) \(\mathrm{J} / \mathrm{kg}...
-
A steel sphere (AISI 1010), \(100 \mathrm{~mm}\) in diameter, is coated with a dielectric material layer of thickness \(2 \mathrm{~mm}\) and thermal conductivity \(0.04 \mathrm{~W} / \mathrm{m} \cdot...

Study smarter with the SolutionInn App


