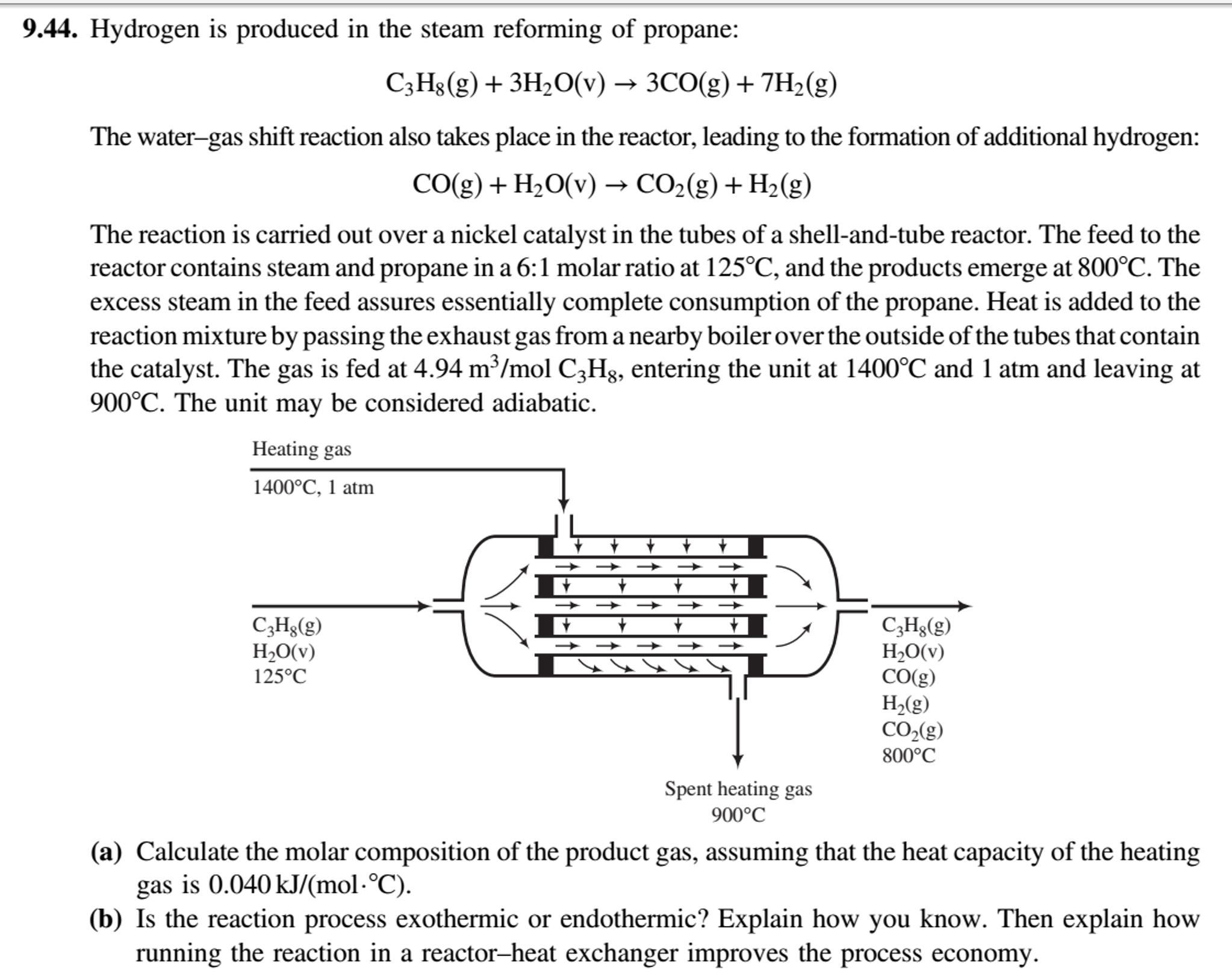
9.44. Hydrogen is produced in the steam reforming of propane: C3H8(g) + 3H2O(v) 3CO(g) +...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
9.44. Hydrogen is produced in the steam reforming of propane: C3H8(g) + 3H2O(v) → 3CO(g) + 7H2(g) The water-gas shift reaction also takes place in the reactor, leading to the formation of additional hydrogen: CO(g) + H2O(v) → CO2(g) + H2(g) The reaction is carried out over a nickel catalyst in the tubes of a shell-and-tube reactor. The feed to the reactor contains steam and propane in a 6:1 molar ratio at 125°C, and the products emerge at 800°C. The excess steam in the feed assures essentially complete consumption of the propane. Heat is added to the reaction mixture by passing the exhaust gas from a nearby boiler over the outside of the tubes that contain the catalyst. The gas is fed at 4.94 m³/mol C3H3, entering the unit at 1400°C and 1 atm and leaving at 900°C. The unit may be considered adiabatic. Heating gas 1400°C, 1 atm C3Hg(g) H2O(v) 125°C C,Hg(g) H,O(v) CO(g) H2(g) CO,(g) 800°C Spent heating gas 900°C (a) Calculate the molar composition of the product gas, assuming that the heat capacity of the heating gas is 0.040 kJ/(mol·°C). (b) Is the reaction process exothermic or endothermic? Explain how you know. Then explain how running the reaction in a reactor–heat exchanger improves the process economy. 9.44. Hydrogen is produced in the steam reforming of propane: C3H8(g) + 3H2O(v) → 3CO(g) + 7H2(g) The water-gas shift reaction also takes place in the reactor, leading to the formation of additional hydrogen: CO(g) + H2O(v) → CO2(g) + H2(g) The reaction is carried out over a nickel catalyst in the tubes of a shell-and-tube reactor. The feed to the reactor contains steam and propane in a 6:1 molar ratio at 125°C, and the products emerge at 800°C. The excess steam in the feed assures essentially complete consumption of the propane. Heat is added to the reaction mixture by passing the exhaust gas from a nearby boiler over the outside of the tubes that contain the catalyst. The gas is fed at 4.94 m³/mol C3H3, entering the unit at 1400°C and 1 atm and leaving at 900°C. The unit may be considered adiabatic. Heating gas 1400°C, 1 atm C3Hg(g) H2O(v) 125°C C,Hg(g) H,O(v) CO(g) H2(g) CO,(g) 800°C Spent heating gas 900°C (a) Calculate the molar composition of the product gas, assuming that the heat capacity of the heating gas is 0.040 kJ/(mol·°C). (b) Is the reaction process exothermic or endothermic? Explain how you know. Then explain how running the reaction in a reactor–heat exchanger improves the process economy.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The following reaction is carried out at 500 K in a container equipped with a movable piston. After the reaction has reached equilibrium, the container has the composition depicted here. Suppose the...
-
If the reaction is carried out at constant V, how does the total pressure change if T increases? H 2 (g) + Cl 2 (g) 2HCl(g) at equilibrium. Assume ideal gas behavior.
-
The following elementary homogeneous gas-phase reaction is carried out isothermally in an ideal, steady state 0.2 L plug flow reactor: 2 F --> P. PThe inlet molar feed consists of a 50%-50% mixture...
-
On May 1, Johnson Corporation purchased inventory for $40,000 on credit. On May 15, Johnson sold inventory with a cost of $10,000 for $25,000 on credit. Prepare journal entries to record these...
-
Why do you think Toyota has come to the point where it is now in crisis due to the braking problem? How can they overcome this problem?
-
Simon Fraser started a landscaping and lawn-care business in April 2012 by investing $20,000 cash in the business in exchange for capital stock. Because his business is in the Midwest, the season...
-
True or False. All practical systems have damping.
-
Scribners Corporation produces fine papers in three production departmentsPulping, Drying, and Finishing. In the Pulping Department, raw materials such as wood fiber and rag cotton are mechanically...
-
Cash $ 20,000 Accounts receivable Refinishing Supplies 5,000 16,000 Tools 65,000 Accumulated Depreciation, tools $ 12,000 Accounts payable 4,000 Mark Madman, capital 4,000 Mark Madman, withdrawals...
-
The following two situations involve the capitalization of borrowing costs. Situation I: On January 1, 2022, Columbia Outfitters signed a fixed-price contract to have Builder Associates construct a...
-
following expenditures related to the vehicle were also made on July 1, 2025: The company pays $2,300 to GEICO for a one-year Insurance policy. The company spends an extra $5,000 to repaint the...
-
A Birr 100, 000 software project is scheduled for 4 weeks. At the end of the third week, the project is 50% complete and the actual costs to date are Birr 90,000. Examine the project health using...
-
What alternate budget-classification systems for the School of Public Affairs at Enormous State University are possible? Identify
-
What happens to WACC as the proportion of debt in a capital structure increases?
-
Sales Depreciation Cost of goods sold Other expenses Interest Cash Accounts receivable Short-term notes payable Long-term debt Net fixed assets Accounts payable Inventory Tax rate Owners' equity 2021...
-
The function in the query that allows me to choose my favorite fields the tables to constitute the query is called as: one: a . Relationship b . Joint c . Projection d . ion
-
Compare Ubuntu (Linux) with CentOS (Linux) and Kali (Linux)? What are the key differences between these Linux distros? In what terms is Ubuntu better than CentOS and Kali?
-
What types of inventory issues Starbucks might reflect upon at the end of each year? The mission of Starbucks is to inspire and nurture the human spiritone person, one cup, and one neighborhood at a...
-
List two applications of freezing-point depression.
-
Why is it generally impossible to predict the rate law for a reaction on the basis of the chemical equation only?
-
Photographers hypo, used to fix negatives during the development process, is sodium thiosulfate pentahydrate. What is the chemical formula of this compound?
-
Last year during the week of July 4th, Murrysville Regional Hospital reported that they treated 264 patients for fireworks-related injuries.
-
The number of stores a person visits on a shopping trip Name and define the four types of measurement levels used in statistics. determine whether the data are qualitative or quantitative.
-
During a recent year, the average U.S. diners spent a month on food prepared outside the home was \($144\) . What is meant by a biased sample? determine whether descriptive or inferential statistics...

Study smarter with the SolutionInn App


