The reaction of iron with oxygen produces hydrogen dioxide (I) according to the following reaction: Fe...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
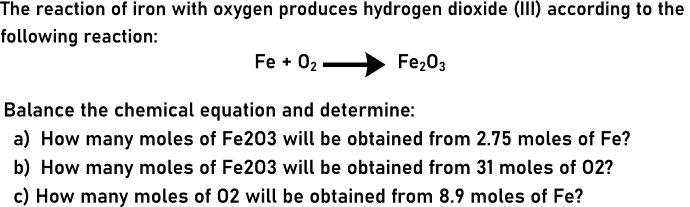
The reaction of iron with oxygen produces hydrogen dioxide (I) according to the following reaction: Fe + 02 Fe203 Balance the chemical equation and determine: a) How many moles of Fe203 will be obtained from 2.75 moles of Fe? b) How many moles of Fe203 will be obtained from 31 moles of 02? c) How many moles of 02 will be obtained from 8.9 moles of Fe? The reaction of iron with oxygen produces hydrogen dioxide (I) according to the following reaction: Fe + 02 Fe203 Balance the chemical equation and determine: a) How many moles of Fe203 will be obtained from 2.75 moles of Fe? b) How many moles of Fe203 will be obtained from 31 moles of 02? c) How many moles of 02 will be obtained from 8.9 moles of Fe?
Expert Answer:
Answer rating: 100% (QA)
Sul2 BFe 02 FeO3 4 By Balancing the chemical eq 2 Fe 15 02 Fe 03 2 male Fe 1 Male of Fe03 I male o... View the full answer

Posted Date:
Students also viewed these chemistry questions
-
How many moles of Al2O3 will be produced when 23.9 g of H2O are reacted according to this chemical equation? 2AlCl3 + 3H2O() ( Al2O3 + 6HCl(g)
-
Write the chemical equation for the reaction of iron metal with hydrochloric acid. Similarly, write the chemical equation for the reaction of copper metal with nitric acid. Discuss the difference...
-
The reaction of iron (II) sulfide in the presence of oxygen produces Fe203 and S02 according to the following reaction: Fes + 02 Fe203 + S02 Balance the chemical equation and determine: a) How many...
-
Gombas Company decided to analyze certain costs for October of the current year. There was no beginning inventory. Units started into production equaled 14000, units transferred out equal 12000, and...
-
Samples of three types of ropes are tested for breaking strength. The data (in pounds) are shown here. At = 0.05, is there a difference in the breaking strength of the ropes? Use the Kruskal-Wallis...
-
PIM Industries, Inc., manufactures electronics components. Each unit costs $30 before the final test. The final test rejects, on average, 5 percent of the 50,000 units manufactured per year. The...
-
After quitting his job at Korn/Ferry, an executive search firm, David Nosal decided to start a competing business. To help get his new business off the ground, Mr. Nosal convinced some of his former...
-
Rio Valde Co. uses a normal cost, job order costing system. In the Mixing Department, overhead is applied using machine hours; in Paving, overhead is applied using direct labor hours. In December...
-
1 Define Management? 2 Define Leadership? 234 4 Explain the Qualities of a successful leader? Define transformational leadership? 5 Explain about Decision making process? Explain the types of...
-
Use information on the endpapers of this book to calculate the average density of the Earth. Where does the value fit among those listed in Tables 1.5 and 14.1? Look up the density of a typical...
-
We wish to estimate the number of nests on a group of 40 islands. The total area of the islands is 100 hectares. a) We sample with probability proportional to size (area) with replacement. (We want a...
-
Current home mortgage interest rates are approximately 4% for a 10 year loan and 3% for a 8 year loan. Which annual payment (A) has a lower present worth (P)?
-
The change in value of securities due to changes in interest rates is known as Blank______.
-
if the current change as a function of time in the equation f ( t ) = 4 t - 3 , find the total charge in the period time from t = 1 s to 6 s
-
Process Improvement Stages: Be specific choosing a area of improvement Please, 1 - Choose a process of improvement in a Warehouse . 2 - Describe the existing process of improvement and choose a...
-
Clothing World is a clothing production company that wants to use a qualitymanagement system to improve efficiency and ensure high-quality production. The leaders at Clothing World meet to discuss...
-
The 2022 accounting records of Crane Transport provide the following information. Payment of interest Cash sales Receipt of dividend revenue Payment of income taxes Net income Payment for merchandise...
-
Explain how the graph of each function can be obtained from the graph of y = 1/x or y = 1/x 2 . Then graph f and give the (a) Domain (b) Range. Determine the largest open intervals of the domain over...
-
Crane l uses 10 kJ of energy to lift a 50 kg box to the roof of a building. Crane 2 uses 20 kl to lift a 100 kg box the same distance. Which crane is more efficient? A. Crane 1 B. Crane 2 C. Both...
-
Christina throws a javelin into the air. As she propels it forward from rest, she does 270 J of work on it. At its highest point, its gravitational potential energy has increased by 70 J. What is the...
-
A runner is moving at a constant speed on level ground. Chemical energy in the runner's body is being transformed into other forms of energy. Most of the chemical energy is transformed into A....

Study smarter with the SolutionInn App

