The reduction potential (E, in V) of MnO4 (aq)/Mn(s) is = 1.68V; E(Mno(s)/Mn2 (aq)) (MnO Given:...
Fantastic news! We've Found the answer you've been seeking!
Question:
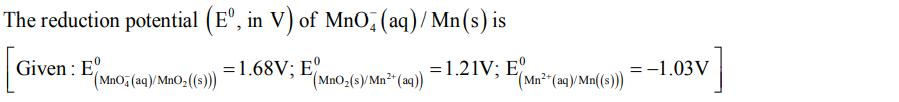
Transcribed Image Text:
The reduction potential (Eº, in V) of MnO4 (aq)/Mn(s) is = 1.68V; E(Mno(s)/Mn2² (aq)) (MnO₂ Given: E (MnO4 (aq)/MnO₂((s))) = 1.21V; E (Mn²+ (aq)/Mn((s))) -1.03V The reduction potential (Eº, in V) of MnO4 (aq)/Mn(s) is = 1.68V; E(Mno(s)/Mn2² (aq)) (MnO₂ Given: E (MnO4 (aq)/MnO₂((s))) = 1.21V; E (Mn²+ (aq)/Mn((s))) -1.03V
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Given that E 5 0.52 V for the reduction Cu+(aq) + e- Cu(s), calculate E, G, and K for the following reaction at 25C: 2Cu+(aq) Cu2+(aq) + Cu(s)
-
The half-cell reduction potential is provided by the Nernst equation (Equation 14-8): (a) On the graph below, plot the reduction potentials for the FADH2/FAD half-cell (E°² = - 0.219 V) when...
-
Given the standard reduction potential for Au3+ in Table 19.1 and Au+(aq) + e- Au(s) E 5 1.69 V answer the following questions. (a) Why does gold not tarnish in air? (b) Will the following...
-
In Problem 10.16, we projected financial statements for Walmart Stores for Years +1 through +5. The data in Chapter 12s Exhibits 12.1612.18 include the actual amounts for 2008 and the projected...
-
1. A risk-neutral consumer is deciding whether to purchase a homogeneous product from one of two firms. One firm produces an unreliable product and the other a reliable product. At the time of the...
-
Use the trial balance presented for Lynch, Inc., in problem (45). Assume that the company will be liquidated and the following transactions will occur: Accounts receivable of $18,000 are collected...
-
2. ETHICS Hector works in Zoey's importing firm. Zoey overhears Hector on the phone say, "O.K., 30,000 ski parkas at $80 per parka. You've got yourself a deal. Thanks a lot." When Hector hangs up,...
-
Tuff Kids Jeans Co. sells blue jeans wholesale to major retailers across the country. Each pair of jeans has a selling price of $ 30 with $ 21 in variable costs of goods sold. The company has fixed...
-
underlying has a current price of $100 annual volatility of 20%. (continuously compounded) risk free rate is 2% p.a. strike price of X = 95 that has 3 months to maturity and cant be exercised ea...
-
The field is less sharply defined. There is heavier use of economics, decision sciences, and behavioral sciences. Identify the branch of accounting described in the quotation.
-
INITIAL TASK (15%): Look closely and examine the sample artwork below (Raphael - The School of Athens-1510-1511) and label the different elements present in the artwork. Note: You could search the...
-
x3+4x2-x+7 is divided by x-5
-
Lucas has a 6-digit number that satisfies the following conditions It reads the same from left to right as from right to left, that is, it is a palindrome is divisible by 9 After crossing out the...
-
A coin has been independently tossed for 1000 times and the number of heads observed has been 432. Find the maximum likelihood estimate of , the probability of observing head in one toss. Find the...
-
Many athletes and cool tech lovers wear a smartwatch, like the Apple Watch, to monitor their fitness and sleep patterns, receive emails, and make phone calls. The watch has an internal battery that...
-
A restaurant purchased a shipment of potatoes for $100 less discounts of 10%, 7%, and 1%. a. Calculate the equivalent discount rate that represents these three discounts. %
-
New Heritage Doll Co. is considering opening five new retail stores in East Coast that would require external financing. Amy Chen, the CFO of New Heritage announced in a board meeting thatthe...
-
Suppose the concentration of glucose inside a cell is 0.1 mm and the cell is suspended in a glucose solution of 0.01 mm. a. What would be the free energy change involved in transporting 10-o mole of...
-
Calculate the energy required to heat 1.00 kg of ethane gas (C2H6) from 25.0oC to 75.0oC first under conditions of constant volume and then at a constant pressure of 2.00 atm. Calculate ÎE,...
-
Under standard conditions, what reaction occurs, if any, when each of the following operations are performed? a. Crystals of I 2 are added to a solution of NaCl. b. Cl 2 gas is bubbled into a...
-
A certain oxide of titanium is 28.31% oxygen by mass and contains a mixture of Ti2+ and Ti3+ ions. Determine the formula of the compound and the relative numbers of Ti2+ and Ti3+ ions.
-
1.5 Financial Times Currency Global Macromaps. The Financial Times provides a very helpful real time global map of currency values and movements online. Use it to track the movements of currency....
-
On May 20, 2016, when the spot rate is \($1.28/,\) a U.S. company buys merchandise from a supplier in Italy, at a cost of 100,000. The spot rate is \($1.25/\) on June 30, the companys year-end....
-
On November 15, 2016, a U.S. company takes delivery of merchandise costing C\($1,000,000\) from a Canadian supplier and records an account payable. On the same date, the company enters a forward...

Study smarter with the SolutionInn App



