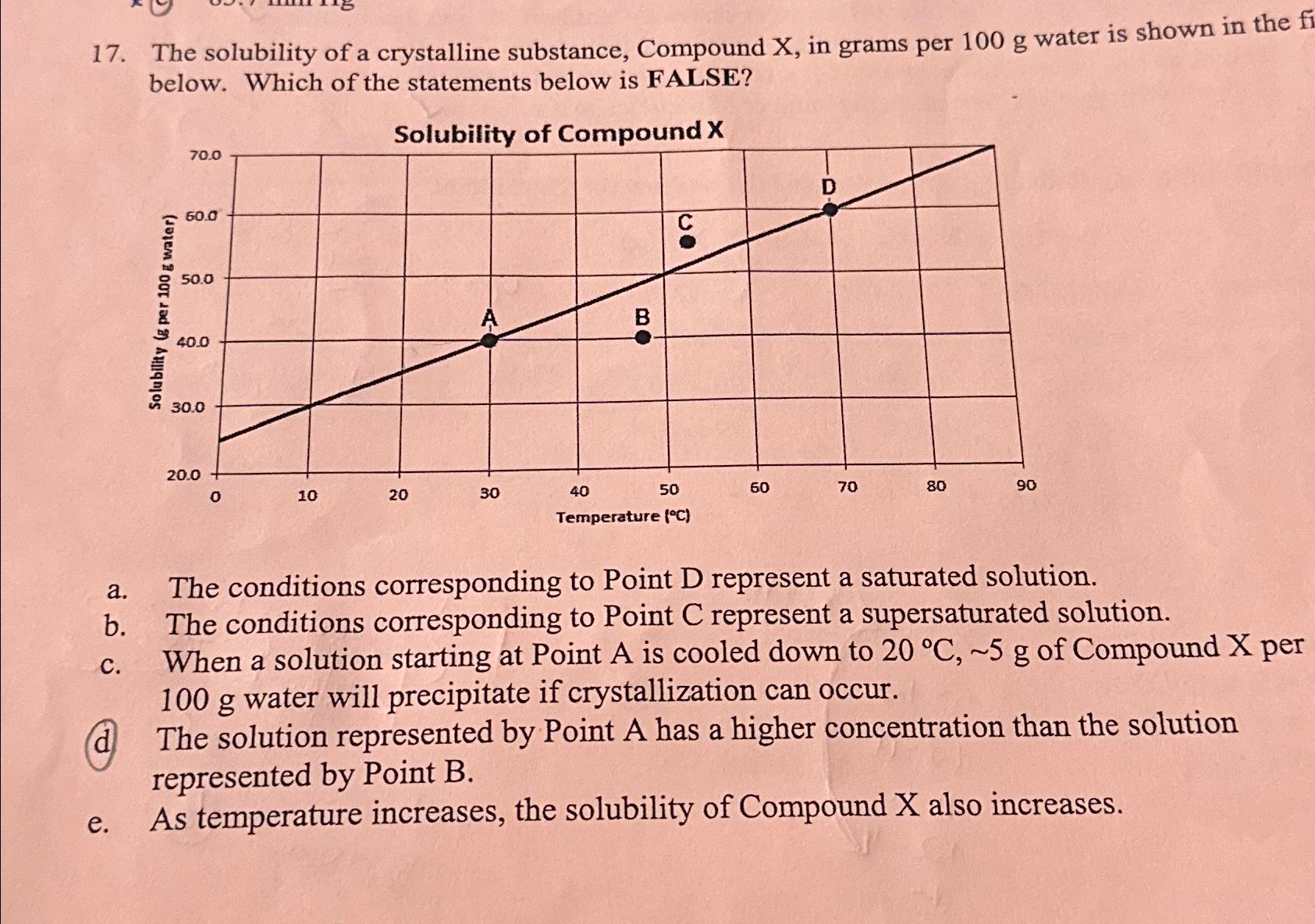
The solubility of a crystalline substance, Compound x , in grams per 100g water is shown in
Fantastic news! We've Found the answer you've been seeking!
Question:
The solubility of a crystalline substance, Compound
x, in grams per
100gwater is shown in the below. Which of the statements below is FALSE?\ Solubility of Compound
x\ a. The conditions corresponding to Point
Drepresent a saturated solution.\ b. The conditions corresponding to Point
Crepresent a supersaturated solution.\ c. When a solution starting at Point
Ais cooled down to
20\\\\deg C,5gof Compound
xper
100gwater will precipitate if crystallization can occur.\ d The solution represented by Point
Ahas a higher concentration than the solution represented by Point
B.\ e. As temperature increases, the solubility of Compound
xalso increases.
Related Book For 

Posted Date:




