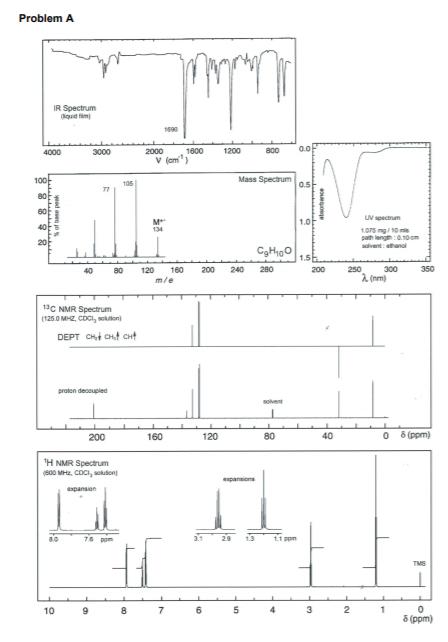
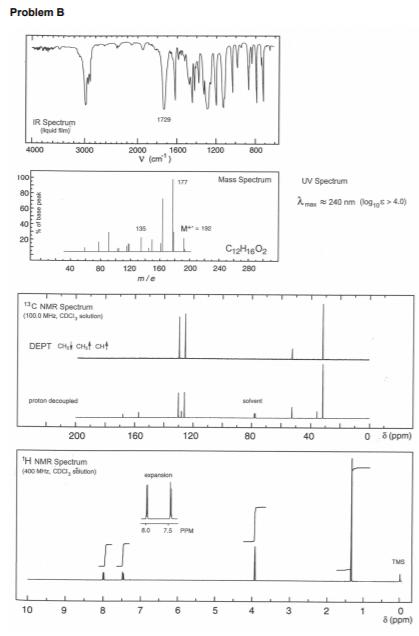
The spectroscopic data and molecular formulas for two compounds (A and B) are given on pages...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
The spectroscopic data and molecular formulas for two compounds (A and B) are given on pages 3 and 7, labelled Problem A and Problem B respectively. Deduce the structures of compounds A and B. As part of your answer you should: (i) Calculate the double bond equivalents (DBE) for each molecule. (ii) Assign the 'H NMR spectra completely, rationalise the number of peaks, relative integral, their multiplicity or splitting pattern where appropriate. (iii) Rationalise the 13C NMR spectra. (iv) Rationalise all numbered peaks in the mass spectra and IR spectra. (v) Provide a brief logical explanation for how you arrived at unambiguous structures for compounds A and B. Problem A IR Spectrum qud tim 1690 4000 3000 2000 1800 1200 a00 0.0 V (am Mass Spectrum 0.5 105 80 60 UV spectnam M 134 1.0 40 1.075 mg/10 i pah leng0 10 om sdivent: ethanal 120 160 m/e 40 80 200 240 280 200 250 300 350 2 (nm) 13C NMR Spectrum (1260 MHZ, CDCI, soulon) DEPT CHt CHt CHt proton decoupled solve 200 160 120 80 40 6 (ppm) H NMR Spectrum (s00 MHz. COCI, sokudon pansions xpansien 76 pom 29 1.3 11 ppm TMS 10 7. 6. 4 5 (ppm) atsobance yad e jo wwww Problem B 1729 IR Spectrum 4000 3000 2000 1600 1200 800 V (cm 100 Mass Spectrum UV Spectrum 177 a0 Aman 240 nm (log,g > 4.0) 135 M 1E 80 120 160 m/e 40 200 240 280 1aC NMR Spectrum (100.0 MR, COci, skton DEPT O CHt aHt proton decouped solrt 200 160 120 80 40 0 5 (ppm) H NMR Spectrum (400 M. CDC, sition) apanskon 7.5 PPM TMS 10 6. 4 3 2. 8 (ppm) yed ose oN The spectroscopic data and molecular formulas for two compounds (A and B) are given on pages 3 and 7, labelled Problem A and Problem B respectively. Deduce the structures of compounds A and B. As part of your answer you should: (i) Calculate the double bond equivalents (DBE) for each molecule. (ii) Assign the 'H NMR spectra completely, rationalise the number of peaks, relative integral, their multiplicity or splitting pattern where appropriate. (iii) Rationalise the 13C NMR spectra. (iv) Rationalise all numbered peaks in the mass spectra and IR spectra. (v) Provide a brief logical explanation for how you arrived at unambiguous structures for compounds A and B. Problem A IR Spectrum qud tim 1690 4000 3000 2000 1800 1200 a00 0.0 V (am Mass Spectrum 0.5 105 80 60 UV spectnam M 134 1.0 40 1.075 mg/10 i pah leng0 10 om sdivent: ethanal 120 160 m/e 40 80 200 240 280 200 250 300 350 2 (nm) 13C NMR Spectrum (1260 MHZ, CDCI, soulon) DEPT CHt CHt CHt proton decoupled solve 200 160 120 80 40 6 (ppm) H NMR Spectrum (s00 MHz. COCI, sokudon pansions xpansien 76 pom 29 1.3 11 ppm TMS 10 7. 6. 4 5 (ppm) atsobance yad e jo wwww Problem B 1729 IR Spectrum 4000 3000 2000 1600 1200 800 V (cm 100 Mass Spectrum UV Spectrum 177 a0 Aman 240 nm (log,g > 4.0) 135 M 1E 80 120 160 m/e 40 200 240 280 1aC NMR Spectrum (100.0 MR, COci, skton DEPT O CHt aHt proton decouped solrt 200 160 120 80 40 0 5 (ppm) H NMR Spectrum (400 M. CDC, sition) apanskon 7.5 PPM TMS 10 6. 4 3 2. 8 (ppm) yed ose oN
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Compounds A and B are isomeric diketones of molecular formula C6H10O2. The 1H NMR spectrum of compound A contains two signals, both singlets, at 2.2 (6 protons) and 2.8 ppm (4 protons). The 1H NMR...
-
Compounds A and B are isomers having the molecular formula C4H8O3. Identify A and B on the basis of their 1H NMR spectra. Compound A: 1.3 ppm (3H, triplet); 3.6 ppm (2H, quartet); 4.1 ppm (2H,...
-
Compounds A and B are carboxylic acids. Identify each one on the basis of its 1H NMR spectrum. (a) Compound A (C3H5ClO2) (Figure 19.10). (b) Compound B (C9H9NO4) has a nitro group attached to an...
-
In 1976, Mohamed EI-Iladad earned an undergraduate accounting degree in his native Egypt. Before he began his accounting career, El-Hadad completed his compulsory service in the Egyptian military...
-
On 28 May 2013 $20 000 cash was stolen from Ming Lee Ltd's night safe. Explain how Ming Lee should account for this event, justifying your answer by reference to relevant Conceptual Framework...
-
Given the following information, what is the optimum portfolio if the lending and borrowing rate is 6%, 8%, or 10%? Assume the Lintner definition of short sales. Covariance with Mean Return Standard...
-
For each of the following situations, calculate the degrees of freedom \((d f)\), identify the critical values (assume \(\mathrm{a}=.05\) [two-tailed]), calculate the \(t\)-statistic \((t)\), make a...
-
Identify the primary audit objectives for a clients year-end discretionary expense accruals. Is it permissible for companies to overstate period-ending expense accruals to make their financial...
-
Payton Corporation will make an investment April 1, 2022. They will receive $9,000 every March 31 for the next six years (2023-2028). If Payton wants to earn 10% on the investment, how much should...
-
How long (max) will it take for the output of a 74ALS00 to go HIGH after its input goes LOW?
-
The marginal product of labor (measured in units of production) for Rotona Corp. is given by MPN = A(200 - N) where A measures productivity and N is the number of labor hours used in...
-
Describe how to calculate a business' payments to suppliers during an accounting period, based on an analysis of its income statement and its beginning and ending balance sheets.
-
The following are several internal control weaknesses of a retail business in regard to its cash payments, accounts payable and inventory: i The inventory of gold jewellery for sale is kept in...
-
What four steps must a business complete to determine whether or not a capital expenditure proposal is acceptable?
-
What is the decision rule for a make-or-buy decision?
-
What is an acceptable accounting rate of return on a capital expenditure proposal?
-
How do power dynamics and hierarchical structures influence the delegation of tasks and responsibilities, and what strategies can leaders employ to cultivate a more democratic and empowering...
-
Doorharmony Company makes doorbells. It has a weighted- average cost of capital of 5% and total assets of $ 5,900,000. Doorharmony has current liabilities of $ 750,000. Its operating income for the...
-
Group the following electron configurations in pairs that would represent similar chemical properties of their atoms: (a) 1s22s22p5 (b) 1s22s1 (c) 1s22s22p6 (d) 1s22s22p63s23p5 (e) 1s22s22p63s23p64s1...
-
The aluminum sulfate hydrate [Al2(SO4)3? xH2O] contains 8.10 percent Al by mass. Calculate x, that is, the number of water molecules associated with each Al2 (SO4)3 unit.
-
Vinyl chloride, H2CPCHCl, undergoes copolymerization with 1,1-dichloroethylene, H2CPCCl2, to form a polymer commercially known as Saran. Draw the structure of the polymer, showing the repeating...
-
Analysis of Canadian Manufacturing Workers Workhours. The time plot in Figure 18 . 22 describes the average annual number of weekly hours spent by Canadian manufacturing workers...
-
Souvenir Sales. The file SouvenirSales.csv contains monthly sales for a souvenir shop at a beach resort town in Queensland, Australia, between 1995 and 2001. [Source: Hyndman and Yang (2018).] Back...
-
Toys R US Revenues. Figure 18 . 23 is a time plot of the quarterly revenues of Toys R US between 1992 and 1995 (thanks to Chris Albright for suggesting the use of these data, which are available in...

Study smarter with the SolutionInn App


