The spin-orbit interaction in an sodium atom is given by H = 21.5, where I and...
Fantastic news! We've Found the answer you've been seeking!
Question:
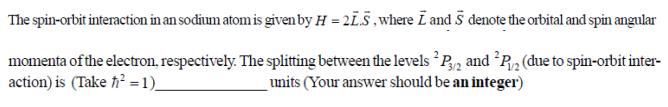
Transcribed Image Text:
The spin-orbit interaction in an sodium atom is given by H = 21.5, where I and 5 denote the orbital and spin angular momenta of the electron, respectively. The splitting between the levels P3/2 and P2 (due to spin-orbit inter- action) is (Take fi = 1)_ units (Your answer should be an integer) The spin-orbit interaction in an sodium atom is given by H = 21.5, where I and 5 denote the orbital and spin angular momenta of the electron, respectively. The splitting between the levels P3/2 and P2 (due to spin-orbit inter- action) is (Take fi = 1)_ units (Your answer should be an integer)
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
An atom in a solid has two energy levels: a ground state of degeneracy g and an excited state of de- generacy 92 at an energy A above the ground state. Show that the partition function Zatom is Zatom...
-
The Zeeman effect is the modification of an atomic spectrum by the application of a strong magnetic field. It arises from the interaction between applied magnetic fields and the magnetic moments due...
-
In stores, boxes should be placed in an organized way otherwise it will be messy. Given a collection of boxes, it is requested to place them on top of each other to reach the minimum possible height....
-
Rosa's employer has instituted a flexible benefits program. Rosa will use the plan to pay for her daughter's dental expenses and other medical expenses that are not covered by health insurance. Rosa...
-
Sora Industries has 60 million outstanding shares, $120 million in debt, $40 million in cash, and the following projected free cash flow for the next four years (see MyFinanceLab for the data in...
-
Repeat Exercise 1.18, but convert to unsigned binary. Data From Problem 18 (a) 4E 16 (b) 7C 16 (c) ED3A 16 (d) 403FB001 16
-
What is a TRR and what is it intended to accomplish?
-
A furniture manufacturer produces two types of tables (country and contemporary) using three types of machines. The time required to produce the tables on each machine is given in the following...
-
This issue was from June of 2022 it involved FMLA for an employee of the Cook county Sheriffs office who was a correctional officer for them. The officer had been working at the Cook county office...
-
Consider the following context-free grammar of expressions E ::= n | (E, E) where n ranges over integers. (a) Present a right-most derivation of the expression ((21, 18), 17). [2 marks] (b) List the...
-
An enzyme has a pH optimum of 7. What will most likely happen if you increase the pH from 7 to 9? The enzyme activity will not be affected. The temperature will decrease. The enzyme activity will...
-
According to the benefit principle of taxation, a businesss tax in a state should be related to the benefits to the business from services provided by the state and local governments. Practically, a...
-
Suppose that a state government levies an ad valorem sales tax on the purchase of all goods at retail but not on the purchases of services. The tax is levied only on final sales of goods and not on...
-
The two most important state taxes are income and general sales taxes, although states also make substantial use of excise taxes, direct business taxes (usually a corporate income tax), and others....
-
In the United States, primary and secondary education is usually provided by local government, although partly financed by state government grants. As a result, there are often substantial...
-
Explain why it may be necessary for state governments to begin to replace unit (per gallon) gasoline excise taxes with an alternative tax or different revenue source. Make the case for a user fee...
-
This company plans to enter a new market next year and asks you to estimate the customer demand in year 2023. You get the past 5-year demand information as below which has been quickly increasing. As...
-
Access the Federation of Tax Administrators Internet site at www. taxadmin.org/state-tax-forms and indicate the titles of the following state tax forms and publications: a. Minnesota Form M-100 b....
-
The refractive index of a compound is 1.622 for 643 nm light. Its density at 20C is 2.99 g cm>'. Calculate the polarizability of the molecule at this wavelength. Take M = 65.5 g mol-1.
-
Sketch the phase diagram for the Mg/Cu system using the following information: Br(Mg) = 648C, Br(Cu) = 1085C; two intermetallic compounds are formed with Br(MgCu2) = 800C and Br(Mg2Cu) = 580C;...
-
R. Viswanathan, R.W. Schmude, Jr., and K.A. Gingerich (J. Phys. Chem. 100,10784 (1996)) studied thermodynamic properties of several boron-silicon gas-phase species experimentally and theoretically....
-
Estimate the depression in the freezing point if \(58.5 \mathrm{~g}\) of \(\mathrm{NaCl}\) is added to \(1 \mathrm{~L}\) of water at atmospheric pressure.
-
Calculate the molal boiling point elevation constant of benzene if its heat of vaporization at \(80.1^{\circ} \mathrm{C}\) is \(30.67 \mathrm{~kJ} / \mathrm{mol}\).
-
A hydrocarbon \(\mathrm{H}_{2}\left(\mathrm{CH}_{2} ight)_{n}\) is dissolved in a solvent \(S\) which freezes at \(9.0^{\circ} \mathrm{C}\). A solution which contains \(0.90 \mathrm{~g}\) of...

Study smarter with the SolutionInn App



