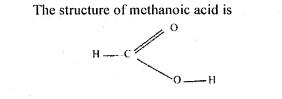
The structure of methanoic acid is L -C -0-H What is the total number of electrons...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
The structure of methanoic acid is L -C -0-H What is the total number of electrons used for bonding in a molecule of methanoic acid? Give reasons. In this question the candidates were required to state the number of electrons used in bonding in a molecule of methanoic acid. The structure of methanoic acid is L -C -0-H What is the total number of electrons used for bonding in a molecule of methanoic acid? Give reasons. In this question the candidates were required to state the number of electrons used in bonding in a molecule of methanoic acid.
Expert Answer:
Answer rating: 100% (QA)
OH Total Number of electrons always depends On total number of bonds and ... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemistry questions
-
What is the total number of electrons that can be held in all orbitals having the same principal quantum number n?
-
Give two reasons why KH(IO3)2 is preferred over benzoic acid as a primary standard for a 0.010 M NaOH solution.
-
What reasons would you give if you were advising someone to study business?
-
What is project scoping? Why is it important to good problem solving?
-
Figure shows an inductance L and a parallel plate capacitor of width w = 20 cm and thickness 0.2 cm. A dielectric with dielectric constant = 4.8 that can completely fill the space between the...
-
As you can see, someone has spilled ink over some of the entries in the balance sheet and income statement of Transylvania Railroad (Table 28.12). Can you use the following information to work out...
-
E Chivers commenced business on 1 January 19X7 and makes his accounts to 31 December every year. For the year ended 31 December 19X7, bad debts written off amounted to 1,200. It was also found...
-
The bills of materials for two finished products (D and E), inventory status, and other relevant information are given below. Compute the planned order releases and projected on-hand balances for...
-
The COSO ERM 2017 framework codifies 20 principles associated with the five components of enterpris Required: Match the following principles with the five components. COSO ERM Principles COSO ERM...
-
SecuriCorp operates a fleet of armored cars that make scheduled pickups and deliveries in the Los Angeles area. The company is implementing an activity-based costing system that has four activity...
-
T) 3 - Chloro- 2- butanol Assign as chiral or achiral. write the asymmetric carbon if chiral. H- OH H- HO- H- -OH H- HO- H2OH U) V) ......... ..........
-
How do we implement change if a College wants to change the learning management system from Moodle to Canvas? What change approach and of the methodology for the project (supported by a diagnosis...
-
1. Who has the access to employee and payroll records aside from the human resources? How does HR protect the privacy of these files? 2. Can someone collect unemployment after being fired in an at...
-
A Bright Shiny Object (BSO) is a set of measures or criteria that aims to add knowledge to what specialists currently emphasize when educating about a discipline (Church, Scrivani, & Paynter, 2019). ...
-
1. Discuss how the original transition into the New York office should have been handled from and International Human Resources Management perspective. 2. Discuss the bank's international...
-
We wish to compare the performance of two different machines: M1 and M2. The following measurements have been made on the machines Program Time on M1 Time on M2 1 10 s 5 s 2 3 s 4 s Which machine is...
-
Toll Road 123 is expected to attract traffic from IH-AB and SH-CD. Use the following data to assess in-scope traffic. 2024 IH-AB 120,000 In Scope Future Growth Lights 108,000 33.3% 2.0% Heavies...
-
A circular concrete shaft liner with Youngs modulus of 3.4 million psi, Poissons ratio of 0.25, unconfined compressive strength 3,500 psi and tensile strength 350 psi is loaded to the verge of...
-
Steve Jackson (age 51) is a single taxpayer living at 3215 Pacific Dr., Del Mar, CA 92014. His Social Security number is 465-88-9415. In 2012, Steve's earnings and income tax withholding as the...
-
Leslie and Leon Lazo are married and file a joint return for 2012. Leslie's Social Security number is 466-47-3311 and Leon's is 467-74-4451. They live at 143 Snapdragon Drive, Reno, NV 82102. For...
-
If Charles, a 16-year-old child model, earns $50,000 a year and is completely self supporting even though he lives with his parents, can his parents claim him as a dependent? Why or why not?...
-
Which statement is false? a. Waste Management made top drawer entries. b. Richard Scrushy of Tyco was never convicted. c. Adelphia used special purpose entities to commit their fraud. d....
-
Which statement is false? a. SASNo. 99 does not prohibit clients from preparing confirmations. b. Enron issued \(\$ 1.2\) billion of stock to an SPE and recorded a \(\$ 1.2\) billion notes...
-
Who is responsible for internal controls? a. External auditors b. Internal auditors c. Management d. Audit Committee e. Board of Directors

Study smarter with the SolutionInn App


