The surface of copper gets tarnished by the formation of copper oxide. N gas was passed...
Fantastic news! We've Found the answer you've been seeking!
Question:
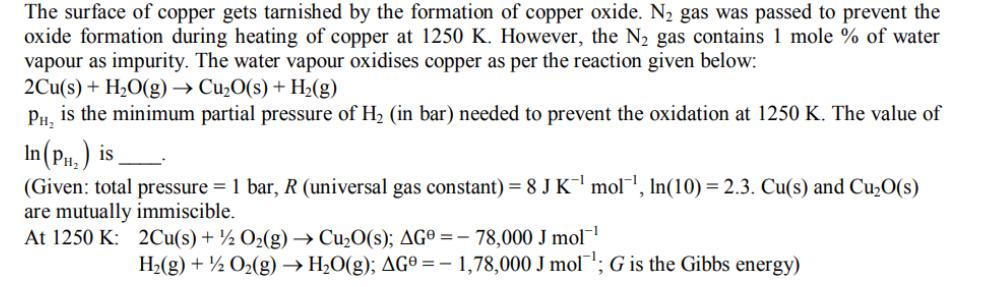
Transcribed Image Text:
The surface of copper gets tarnished by the formation of copper oxide. N gas was passed to prevent the oxide formation during heating of copper at 1250 K. However, the N gas contains 1 mole % of water vapour as impurity. The water vapour oxidises copper as per the reaction given below: 2Cu(s) + HO(g) CuO(s) + H(g) PH is the minimum partial pressure of H (in bar) needed to prevent the oxidation at 1250 K. The value of In (P) is (Given: total pressure = 1 bar, R (universal gas constant) = 8 J K mol, In(10) = 2.3. Cu(s) and CuO(s) are mutually immiscible. At 1250 K: 2Cu(s) + O(g) CuO(s); AG-78,000 J mol- H(g) + 1/2O(g) HO(g); AG = -1,78,000 J mol G is the Gibbs energy) The surface of copper gets tarnished by the formation of copper oxide. N gas was passed to prevent the oxide formation during heating of copper at 1250 K. However, the N gas contains 1 mole % of water vapour as impurity. The water vapour oxidises copper as per the reaction given below: 2Cu(s) + HO(g) CuO(s) + H(g) PH is the minimum partial pressure of H (in bar) needed to prevent the oxidation at 1250 K. The value of In (P) is (Given: total pressure = 1 bar, R (universal gas constant) = 8 J K mol, In(10) = 2.3. Cu(s) and CuO(s) are mutually immiscible. At 1250 K: 2Cu(s) + O(g) CuO(s); AG-78,000 J mol- H(g) + 1/2O(g) HO(g); AG = -1,78,000 J mol G is the Gibbs energy)
Expert Answer:
Related Book For 

Fundamentals Of Momentum Heat And Mass Transfer
ISBN: 9781118947463
6th Edition
Authors: James Welty, Gregory L. Rorrer, David G. Foster
Posted Date:
Students also viewed these chemistry questions
-
The vapour pressure of pure liquid A at 293 K is 68.8 kPa and that of pure liquid B is 82.1 kPa. These two compounds form ideal liquid and gaseous mixtures. Consider the equilibrium composition of a...
-
At 10 000 K the ionization reaction for Ar is: Ar Ar+ + e with equilibrium constant of K = 4.2 104. What should the pressure be for a mole concentration of argon ions (Ar+) of 10%?
-
At 10 000 K the two ionization reactions for N and Ar as have equilibrium constants of K1 = 4.2 104 and K2 = 6.3 104, respectively. If we start out with 1 kmol Ar and 0.5 kmol N2, what is the...
-
PWX Inc. has the following information for its years ended June 30: Required: Calculate the accounts receivable turnover and average collection period for 20X3 and 20X2. Comment on the trend. What...
-
Malic acid, C4H6O5, has been isolated from apples. Because this compound reacts with 2 molar equivalents of base, it is a dicarboxylic acid. (a) Draw at least five possible structures. (b) If malic...
-
Data for Research Enterprises follows: Compute the dollar amount of change and the percentage of change in Research Enterprises's working capital each year during 2019 and 2018. What do the...
-
The ____________ decision model views individuals as making optimizing decisions, whereas the ____________ decision model views them as making satisficing decisions. (a) behavioral/judgmental...
-
In 1895, the first U.S. Open Golf Championship was held. The winner's prize money was $150. In 2006, the winner's check was $1,225,000. What was the annual percentage increase in the winner's check...
-
03 Question (6 points) Although there are clear benefits to free trade, governments impose tariffs to protect domestic industries. The figure below illustrates the effects of a per-unit tariff placed...
-
Beth R. Jordan lives at 2322 Skyview Road, Mesa, AZ 85201. She is a tax accountant with Mesa Manufacturing Company, 1203 Western Avenue, Mesa, AZ 85201 (employer identification number 11-1111111)....
-
Quand un chercheur risque-t-il une erreur de type II ? a) tout moment H1, est rejet b) tout moment H0, est rejet c) chaque fois que la dcision est "ne pas rejeter H0" d) Aucune des autres options...
-
Nine years ago, Ann entered into a $34,000 mortgage loan of 20 years and at a fixed rate of 12% p.a. (compounding monthly) with HSBC. a. What is Ann's monthly payment? b. How much does she still owe...
-
Information for two alternative projects involving machinery investments follows. Project 1 requires an initial investment of $ 2 5 6 , 5 0 0 . Project 2 requires an initial investment of $ 1 7 4 , 0...
-
If the average return on common stocks was 13.3%, the average Treasury bill rate was 3.8%, and the average inflation rate was 3.2% what would be the expected nominal and approximate real market...
-
Problem #4: As part of a promotion, you can buy a new living room set today and make no payments for 3 years. Starting exactly 3 years from today, you will make 5 annual payments of $1500 each. If...
-
1 - The management of KADJI expects to receive cash revenues of $120m, $180m and $300m four, five and six years from now, respectively.KADJI can earn an annual return of 5%, compounded annually, on...
-
Thank you in advanced. Will thumbs up immediately Cobalt Industries had sales of 154,600 units at a price of $9.57 per unit. It faced fixed operating costs of $241,000 and variable operating costs of...
-
A genetically engineered strain of Escherichia coli (E. coli) is used to synthesize human insulin for people suffering from type I diabetes mellitus. In the following simplified reaction scheme,...
-
During the development of a 300-ft ship, it is desired to test a 10% scale model in a towing tank to determine the drag characteristics of the hull. Determine how the model is to be tested if the...
-
The mass transfer device shown in the figure at the top of the next column is used to carry out the controlled release of a vapor-phase pheromone drug used in pest control. The solid drug sublimes at...
-
In a velocity field where v = 400[(y/L) 2 e x + (x/L) 2 e y ] fps, determine the pressure gradient at the point (L, 2L). The y axis is vertical, the density is 64.4 Ib m /ft 3 and the flow may be...
-
Distinguish among the three types of responsibility
-
Using the information in E7-1, assume that in July 2002, Voss Company incurs the following manufacturing overhead costs. Instructions (a) Prepare a flexible budget performance report, assuming that...
-
Samano Company uses flexible budgets to control its selling expenses. Monthly sales are expected to range from \($170,000\) to \($200,000\). Variable costs and their percentage relationship to sales...

Study smarter with the SolutionInn App



