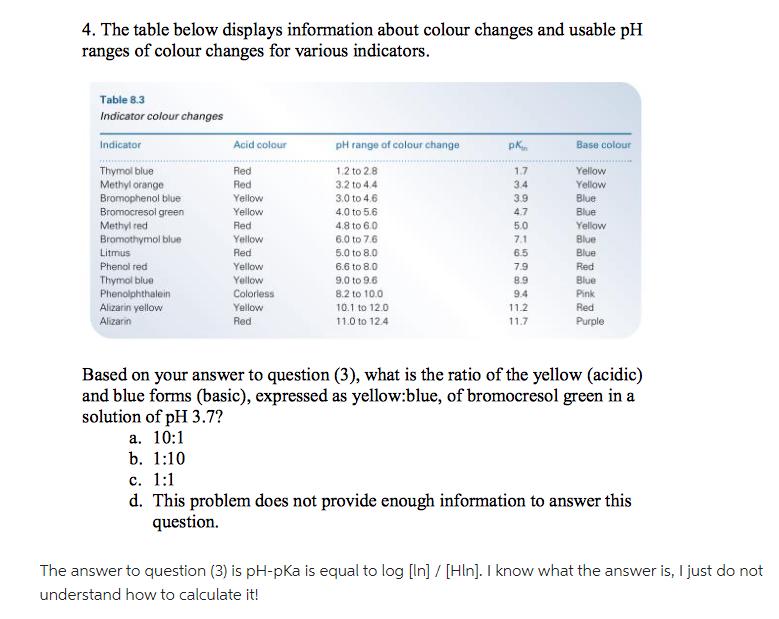
The table below displays information about colour changes and usable pH ranges of colour changes for...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The table below displays information about colour changes and usable pH ranges of colour changes for various indicators. Table 8.3 Indicator colour changes Indicator Thymol blue Methyl orange Bromophenol blue Bromocresol green Methyl red Bromothymol blue Litmus Phenol red Thymol blue Phenolphthalein Alizarin yellow Alizarin Acid colour Red Red Yellow Yellow Red Yellow Red Yellow Yellow Colorless Yellow Red pH range of colour change 1,2 to 2.8 3.2 to 4.4 3.0 to 4.6 4.0 to 5.6 4.8 to 6.0 6.0 to 7.6 5.0 to 8.0 6.6 to 8.0 9.0 to 9.6 8.2 to 10.0 10.1 to 12.0 11.0 to 12.4 pk 1.7 3.4 3.9 4.7 5.0 7.1 76 6.5 7.9 8.9 9.4 11.2 11.7 Base colour Yellow Yellow Blue Blue Yellow Blue Blue Red Blue Pink Red Purple Based on your answer to question (3), what is the ratio of the yellow (acidic) and blue forms (basic), expressed as yellow:blue, of bromocresol green in a solution of pH 3.7? a. 10:1 b. 1:10 c. 1:1 d. This problem does not provide enough information to answer this question. The answer to question (3) is pH-pKa is equal to log [In] / [Hin]. I know what the answer is, I just do not understand how to calculate it! The table below displays information about colour changes and usable pH ranges of colour changes for various indicators. Table 8.3 Indicator colour changes Indicator Thymol blue Methyl orange Bromophenol blue Bromocresol green Methyl red Bromothymol blue Litmus Phenol red Thymol blue Phenolphthalein Alizarin yellow Alizarin Acid colour Red Red Yellow Yellow Red Yellow Red Yellow Yellow Colorless Yellow Red pH range of colour change 1,2 to 2.8 3.2 to 4.4 3.0 to 4.6 4.0 to 5.6 4.8 to 6.0 6.0 to 7.6 5.0 to 8.0 6.6 to 8.0 9.0 to 9.6 8.2 to 10.0 10.1 to 12.0 11.0 to 12.4 pk 1.7 3.4 3.9 4.7 5.0 7.1 76 6.5 7.9 8.9 9.4 11.2 11.7 Base colour Yellow Yellow Blue Blue Yellow Blue Blue Red Blue Pink Red Purple Based on your answer to question (3), what is the ratio of the yellow (acidic) and blue forms (basic), expressed as yellow:blue, of bromocresol green in a solution of pH 3.7? a. 10:1 b. 1:10 c. 1:1 d. This problem does not provide enough information to answer this question. The answer to question (3) is pH-pKa is equal to log [In] / [Hin]. I know what the answer is, I just do not understand how to calculate it!
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Based on your answer to question 4, should Mesa consider hedging its exchange rate risk? If so, explain how it could hedge using forward contracts, futures contracts, and currency options.
-
Based on your answer to Question 5-17, if Pennsylvania's government aims to correct the steel market's negative externality via an effluent fee, is the appropriate fee higher or lower now that steel...
-
Based on your answer to question 9, construct a first-quarter 2015cash flow forecast for Westmark Industrial. Westmark Industrials annual income statement and balance sheet for December 31, 2014...
-
Show that in the limit ??x ?? 0 and ??t ?? 0, the difference Equation (3.12) is equivalent to the differential Equation (2.5). GIVENThe difference equation for one-dimensional transient...
-
Lovers of classical music persuade Congress to impose a price ceiling of $40 per concert ticket. As a result of this policy, do more or fewer people attend classical music concerts?
-
Evaluate the following integrals. sin (In x) 4x
-
Why do we need to gather requirements?
-
How should governments report capital projects and debt service activities in their government wide statements?
-
Labor Supply Define or explain the terms and what they mean to labor supply. Give examples where applicable. A. Utility maximization, preferences, and budget constraint B. Income and substitution...
-
Write stubs for all the constructors and methods in the Student class of Figures 8.2a and 8.2b. Each stub should print the constructor or method name followed by the passed-in values of all...
-
Using MatLab, Simulate BPSK, QPSK, QAM16, QAM64 and StarQAM16 and plot the SER vs SNR curves on the same axes. For each modulation scheme the SER rapidly reduces at a particular SNR. Why does this...
-
A pair of dice is rolled. What is the probability that the resulting sum is eight?
-
A researcher chooses three leaves from a target environment and classifies each sample as fungus free or contaminated. Suppose that a leaf has a probability of 0.2 of being infected. In Problems...
-
Suppose the National League team has a probability of \(\frac{3}{5}\) of winning a World Series game and the American League team has a probability of \(\frac{2}{5}\). The series is over as soon as...
-
Fill in the blank field in this text: While in the Door tool you can change the door style and size via the [1]_________________________________within the Properties Palette.
-
Fill in the blank field in this text: Clicking on the[1]_________________________ next to the Zoom icon will list additional zoom tools not currently shown in the View toolbar.
-
Analyze the best ways for UAE businesses to implement artificial intelligence (AI) ethics. Which codes, concepts, and practices governing ethical AI must be created by UAE organizations (both private...
-
One Way Cellular accountants have assembled the following data for the year ended September 30, 2014: Prepare the operating activities section using the indirect method for One Way Cellulars...
-
As the inside radius of an open tubular column is decreased, the maximum possible column efficiency increases and sample capacity decreases. For a thin stationary phase that equilibrates rapidly with...
-
Fill in the blanks: (a) The product of a reaction between a Lewis acid and a Lewis base is called ________. (b) The bond between a Lewis acid and a Lewis base is called ________ or _______. (c)...
-
Combustion analysis of an organic compound gave the composition 71.17 0.41 wt% C, 6.76 0.12 wt% H, and 10.34 0.08 wt% N. Find the coefficients h and n and their uncertainties x and y in the...
-
Classify these Bluetooth layers using the OSI model: a. baseband; b. L2CAP; c. RFCOMM.
-
Use the power state machine of Fig. 8.12 to determine the energy used in these use cases: a. idle 1 s; receive 10 ms; idle 0.1 s; transmit 5 s; b. sleep 1 min; receive 50 ms; idle 0.1 s; receive 100...
-
What are the main phases of a design review?

Study smarter with the SolutionInn App



