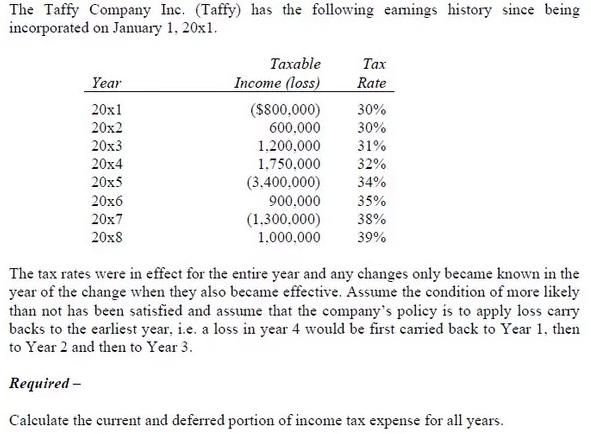
The Taffy Company Inc. (Taffy) has the following eamings history since being incorporated on January 1,...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The Taffy Company Inc. (Taffy) has the following eamings history since being incorporated on January 1, 20x1. Year 20x1 20x2 20x3 20x4 20x5 20x6 20x7 20x8 Taxable Income (loss) ($800.000) 30% 600.000 30% 31% 32% 34% 1.200,000 1,750,000 (3.400,000) 900,000 Tax Rate (1.300.000) 1,000,000 35% 38% 39% The tax rates were in effect for the entire year and any changes only became known in the year of the change when they also became effective. Assume the condition of more likely than not has been satisfied and assume that the company's policy is to apply loss carry backs to the earliest year, i.e. a loss in year 4 would be first carried back to Year 1, then to Year 2 and then to Year 3. Required - Calculate the current and deferred portion of income tax expense for all years. The Taffy Company Inc. (Taffy) has the following eamings history since being incorporated on January 1, 20x1. Year 20x1 20x2 20x3 20x4 20x5 20x6 20x7 20x8 Taxable Income (loss) ($800.000) 30% 600.000 30% 31% 32% 34% 1.200,000 1,750,000 (3.400,000) 900,000 Tax Rate (1.300.000) 1,000,000 35% 38% 39% The tax rates were in effect for the entire year and any changes only became known in the year of the change when they also became effective. Assume the condition of more likely than not has been satisfied and assume that the company's policy is to apply loss carry backs to the earliest year, i.e. a loss in year 4 would be first carried back to Year 1, then to Year 2 and then to Year 3. Required - Calculate the current and deferred portion of income tax expense for all years.
Expert Answer:
Answer rating: 100% (QA)
Calculate the current and deferred portion of income tax expense for each year Year 20x1 Taxable Inc... View the full answer

Related Book For 

Financial Reporting And Analysis
ISBN: 9781260247848
8th Edition
Authors: Lawrence Revsine, Daniel Collins, Bruce Johnson, Fred Mittelstaedt, Leonard Soffer
Posted Date:
Students also viewed these accounting questions
-
Salem Corporation designs and manufactures wind turbine power generating systems and sells them to private energy producers. Many of their sales are structured to allow their customers to pay for...
-
Peter's Pewter sells figurines. During the month of August 2022, the following transactions occurred. Aug 3 Peter invested $4,000 into his business Aug 7 Aug 16 Sold inventory to Joyce Fontane for...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
An LNC can detect alterations in the record by Select one: a. Examining notes written in the margin b. Looking through late entries for detailed explanations c. Finding a doctor's explanation that is...
-
A path cover of a directed graph G = (V, E) is a set P of vertex-disjoint paths such that every vertex in V is included in exactly one path in P. Paths may start and end anywhere, and they may be of...
-
Take a control volume around your kitchen refrigerator and indicate where the components shown in Figure 1.6 are located and show all flows of energy transfer.
-
How to oppose a motion for summary judgment?
-
Each autumn, as a hobby, Pauline Spahr weaves cotton placemats to sell at a local craft shop. The mats sell for $ 20 per set of four mats. The shop charges a 10% commission and remits the net...
-
A 39.0 g particle is released from rest at x = A. What is the speed of the particle at x = C? Assume mechanical energy is conserved. U (J) 6 S 3 2 1 A B x C D
-
Your company is competing for business in the toy robot industry Prepare a sales, production, direct materials, and direct labor budget based on the following assumptions Assumptions Q1 Q2 Q4 1....
-
On January 1, 2021, Puma Company signs a 10-year agreement to lease a standard non-specialized storage building from Reebok, Inc. The following information pertains to this lease agreement: The...
-
2 Factor: x3 + 5x - 2x - 10.
-
A soft tennis ball is dropped onto a hard floor from a height of 1.55 m and rebounds to a height of 1.12m. (Assume that the positive direction is upward.) (a) Calculate its velocity (in m/s) just...
-
Great Furniture Inc. (GF) manufactures a variety of furniture for household use and just two items for office use: desks and cabinets. The production process for desks and cabinets is similar,...
-
Suppose a firm has had the following historic sales figures. Year Sales ($) 2016 1,470,000 2017 1,700,000 2018 1,600,000 2019 2,170,000 2020 1,760,000 What would be the forecast for next...
-
Implement the following pseudo-code in ARM assembly. R5 = 0 R3=0x0000AA00 R0= 70 R1 = 21 while R0 R1 do R5 = (R5 + R3) < < 2 R1 = R1 +3 end while
-
What are CRISPR-Cas9 and its limitations?
-
Dr. Chan obtained a $15,000 demand loan at prime plus 1.5% on September 13 from the Bank of Montreal to purchase a new dental X-ray machine. Fixed payments of $700 will be deducted from the dentists...
-
On November 1, 20X1, Gerakos Corporation sold software and a six-month technical support contract to a customer for $80,000. Gerakos sells the same software without technical support for $60,000. It...
-
Jones Corporation switched from the LIFO method of costing inventories to the FIFO method at the beginning of 20X1. The LIFO inventory at the end of 20X0 would have been $80,000 higher using FIFO....
-
Union Company acquired machinery on January 2, 20X1, for $315,000. The machinerys estimated useful life is 10 years, and the estimated residual value is $15,000. Union estimates that the machine will...
-
Show that \[ \gamma_{i}=\frac{\bar{\phi}_{i}}{\phi_{i}} \]
-
Carbon dioxide occupies a tank at \(100^{\circ} \mathrm{C}\). If the volume of the tank is \(0.5 \mathrm{~m}^{3}\) and the pressure is \(500 \mathrm{kPa}\), determine the mass of the gas in the tank.
-
If a cylinder of volume \(0.1 \mathrm{~m}^{3}\) is filled with \(1.373 \mathrm{~kg}\) of ammonia at \(1.95 \mathrm{MPa}\), determine the temperature at which ammonia exists in the cylinder. Assume...

Study smarter with the SolutionInn App


