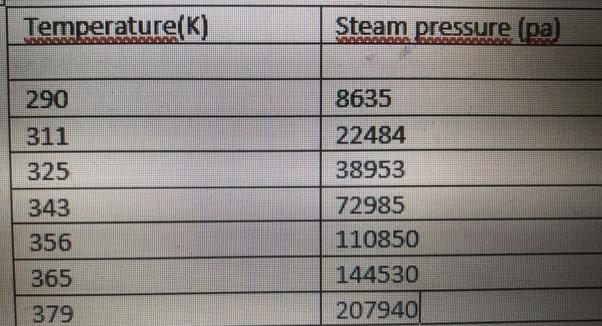
The vapor pressure of the benzene at different temperatures is given in the table on the...
Fantastic news! We've Found the answer you've been seeking!
Transcribed Image Text:
The vapor pressure of the benzene at different temperatures is given in the table on the side. Match this Clapeyron equation (linear model). Clapeyron equation: log P = A + B/T Here T is absolute temperature (K), P pressure (Pa), A and B are parameters of equation. a. (15 points) Calculate the parameters of equation and coefficient of determination (R2) and create and write the results in a table similar to the following (Perform your calculations here manually) (10 points) Show the measured (given in the table) values and your linear model in the same chart (You can use Excel here Temperature(K) Steam pressure (pa) 290 8635 311 22484 325 38953 343 72985 110850 144530 207940 356 365 379 The vapor pressure of the benzene at different temperatures is given in the table on the side. Match this Clapeyron equation (linear model). Clapeyron equation: log P = A + B/T Here T is absolute temperature (K), P pressure (Pa), A and B are parameters of equation. a. (15 points) Calculate the parameters of equation and coefficient of determination (R2) and create and write the results in a table similar to the following (Perform your calculations here manually) (10 points) Show the measured (given in the table) values and your linear model in the same chart (You can use Excel here Temperature(K) Steam pressure (pa) 290 8635 311 22484 325 38953 343 72985 110850 144530 207940 356 365 379
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The following table gives the vapor pressure of hexafluorobenzene (C6F6) as a function of temperature: (a) By plotting these data in a suitable fashion, determine whether the Clausius-Clapeyron...
-
The Dew Point The vapor pressure of water (see Problem 18.88) decreases as the temperature decreases. If the amount of water vapor in the air is kept constant as the air is cooled, a temperature is...
-
At 300C, the vapor pressure of Hg is 32.97 torr. What mass of Au would have to be dissolved in 5.00 g of Hg to lower its vapor pressure to 25.00 torr?
-
A table of properties for methyl chloride lists the saturation pressure as 116.7 psia at 100oF. At 100oF, this table also lists hfg = 154.85 Btu/lbm, and vfg = 0.86332 ft3/lbm. Estimate the...
-
Compare the annual life insurance premium of Garrett Townse who is 30 years old, and purchases a $100,000, 10-year level term policy using a non-tobacco rate to the premium paid by Edward Collins,...
-
Consider an exchange economy with two goods, two agents and two possible states of the world. The preference relation of with \(x_{n}^{i}(\omega)\) denoting the quantity of good \(n=1,2\) consumed by...
-
To the left of z = 0.35 Find the indicated area under the standard normal curve. If convenient, use technology to find the area.
-
A firms current profits are $550,000. These profits are expected to grow indefinitely at a constant annual rate of 5 percent. If the firms opportunity cost of funds is 8 percent, determine the value...
-
Please explain why the decision is administrative in character and how is it made under an enactment. The statute is below and the made up case study is after the statute (also made up). PART 2...
-
On August 31, Sunland Marine had an inventory of 21 boats at a cost of $2,100 each. Sunland does not expect any returns from sales of boats. The company uses a perpetual inventory system. During...
-
How do I create a flow chart for the following steps I came up with (are they correct)? I am having an issue conceptualizing the flow chart for these steps: 1). Connect timecard/ contract with a...
-
A biorefinery produces 94 million gallons of ethanol per year from corn grain. It can produce 2.9 gallons of ethanol from every bushel. How many acres of corn (assuming an average yield of 173...
-
" In her 2 0 1 2 chapter on Critical Thinking and Reasoning, Terri Russ defines critical thinking as 'active thinking in which we evaluate and analyze information in order to determine the action. '...
-
Find all the "daffodil numbers" between 100 and 999 and output them. "Daffodil number" refers to a three-digit number, and the cube of each digit is exactly equal to the number itself. For example,...
-
The following data refers to the results of the inspection of shirts in a factory. No. of Shirts No. of Imperfections Bundle No. 1 1234 2 4 5 15 15 15 15 15 5 3 10 8 18 The number of imperfections in...
-
Assume that P = Tshs 1 0 0 , 0 0 0 , i = 1 2 % , and n = 3 years. Find FV when interest is compounded: ( a ) annually
-
The figure below shows a circuit with a 12.0-V battery connected to four resistors. How much power is delivered to each resistor? (Consider the following values: R = 1.30 Q, R = 1.95 02, R3 = 4.200,...
-
The first law of thermodynamics is sometimes whimsically stated as, You cant get something for nothing, and the second law as, You cant even break even. Explain how these statements could be...
-
Initially a mixture contains 0.795 mol each of N2 and O2 in an 8.00-L vessel. Find the composition of the mixture when equilibrium is reached at 3900oC. The reaction is and Kc = 0.0123 at 3900oC....
-
Describe the physical characteristics of white phosphorus. Is it found in any modern matches? Why or why not?
-
The nucleus is a metastable nucleus of technetium-99; it is used in medical diagnostic work. Technetium 99m decays by emitting gamma rays. A 2.5-μg (microgram) sample has an activity of...
-
Is an oscillating object in translational equilibrium?
-
How far above Farth's surface must a \(10,000-\mathrm{kg}\) boulder be moved to increase the mass of the Earthboulder system by \(2.50 \mathrm{mg}\) ? Assume the same ratio of energy change to mass...
-
Show that for small displacements the restoring force exerted on part 2 of the displaced string in Figure 15.14 is linearly proportional to the displacement of that part from its equilibrium...

Study smarter with the SolutionInn App