The volume occupied by an atom is greater than the volume of the nucleus by a...
Fantastic news! We've Found the answer you've been seeking!
Question:
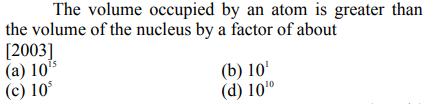
Transcribed Image Text:
The volume occupied by an atom is greater than the volume of the nucleus by a factor of about [2003] (a) 105 (c) 10 (b) 10 (d) 100 The volume occupied by an atom is greater than the volume of the nucleus by a factor of about [2003] (a) 105 (c) 10 (b) 10 (d) 100
Expert Answer:
Related Book For 

Thermodynamics for Engineers
ISBN: ?978-1133112860
1st edition
Authors: Kenneth A. Kroos, Merle C. Potter
Posted Date:
Students also viewed these physics questions
-
The volume occupied by a gas depends linearly on degrees Celsius at constant pressure, but it is not directly proportional to degrees Celsius. However, it is directly proportional to kelvins. What is...
-
The volume occupied by 20 kg of water at 300 C and 10 MPa is nearest: (A) 0.028 m3 (B) 0.088 m3 (C) 0.22 m3 (D) 0.36 m3
-
The volume occupied by one gram-equivalent of oxygen at 273 K and 550 mmHg is A) 7.74 L B) 5.6 L C) 6.84 L D) 6.08 L
-
Briar Company manufactures and sells dresses at a variable cost of $32 each and a fixed cost of x. It can sell 6,600 dresses at a selling price of $60 to earn an operating income of $14,800 (Option...
-
To underscore WFMs relatively high prices, some skeptics add Whole Paycheck to the companys motto of Whole Foods, Whole People whole Planet; they also point out that, despite discounts of 15 percent...
-
List the evaluative criteria and the importance of each that you would use in purchasing (or renting or giving to) the options in Question 23. Would situational factors change the criteria? The...
-
Explain why some production costs must be allocated rather than traced to products. Name several such costs. Would such costs be classified as direct or as indirect costs?
-
Lorien Technologies, Inc., purchased the following securities during 2011: The following transactions occurred during 2012: a. On January 1, 2012, Lorien purchased Security E for $12,000. Security E...
-
Solve the following triangle. Round answers to the nearest hundredth M 51 37 m
-
The proposed rates were not in the range the CEO expected given the pricing analysis. The CEO has asked the pricing actuary to verify the total projected loss cost excluding potential large storm...
-
What is a sequence of transformation to describe the figures in the graph? 10F 8 C B B A A 864 2 2 4 6 8 10 2 Rotate 90 clockwise about the origin, then Reflect over the y-axis Rotate 270...
-
What is the impact of organizational justice on employee satisfaction and retention, and how can companies design fair and transparent processes for performance evaluation and career advancement ?
-
How do socio-cultural factors embedded within organizational contexts shape employee perceptions of fairness and equity in reward allocation systems ?
-
On January 1, 2021, you deposited $6,600 in a savings account. The account will earn 9 percent annual compound interest, which will be added to the fund balance at the end of each year. Required: 1....
-
discuss the paradoxical nature of organizational structure in balancing the need for stability and agility in rapidly evolving markets ?
-
A 25% rise is predicted for the demand in 2020 based on the annual output of 944 units in 2019. Quarter relatives are Q1 = 1.1; Q2= 0.6; Q3= 1.6; and Q4 = 0.8. What is the forecast for the third...
-
On 1/1/2020, Co. X acquired 80% of the voting stock of Co. Y at a cost of $150,000. The book values of Co. Y's assets and liabilities at the date of acquisition were $200,000 and $30,000,...
-
If the joint cost function for two products is C(x, y) = xy2 + 1 dollars (a) Find the marginal cost (function) with respect to x. (b) Find the marginal cost with respect to y.
-
Air is heated from 20 C to 800 C at constant pressure of 200 kPa in a cylinder with an initial volume of 4000 cm3. The entropy change, assuming an ideal gas with constant specific heats, is nearest:...
-
Superheated steam enters an insulated turbine at 20 MPa and 600 C at a mass flow rate of 1000 kg/min. If the steam leaves the turbine at a) 10 kPa, b) 40 kPa, c) 80 kPa as a saturated vapor, what are...
-
Air at 700C is expanded in an insulated cylinder such that the volume increases by a factor of 8. Estimate the final temperature, assuming a quasi-equilibrium process. Also, calculate the work...
-
Lomax Companys Statement No. 69 disclosures included the following information: REQUIRED: Using the information for Lomax Company in problems 13, 14, and 15 and in this problem: a. Compute the value...
-
Lomax Company reported the following expenses in its financial statements (in thousands): REQUIRED: Using the reserve disclosure for Lomax Company given in problem 13 and the data presented in this...
-
Discuss the ratios computed for Lomax Company in problems 13, 14, 15, and 16. What is your assessment of the performance and future potential of Lomax Company? Problems 13 The following reserve table...

Study smarter with the SolutionInn App



