The weight percentage of hydrogen in Q, formed in the following reaction sequence, is CI 1....
Fantastic news! We've Found the answer you've been seeking!
Question:
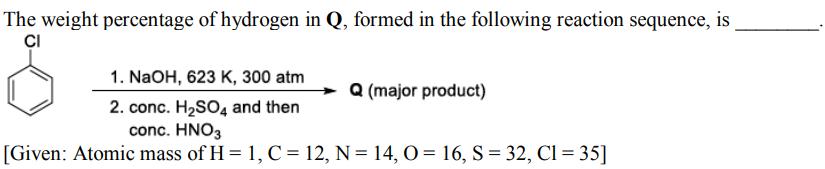
Transcribed Image Text:
The weight percentage of hydrogen in Q, formed in the following reaction sequence, is CI 1. NaOH, 623 K, 300 atm 2. conc. H₂SO4 and then conc. HNO3 [Given: Atomic mass of H = 1, C = 12, N = 14, 0= 16, S = 32, Cl = 35] Q (major product) The weight percentage of hydrogen in Q, formed in the following reaction sequence, is CI 1. NaOH, 623 K, 300 atm 2. conc. H₂SO4 and then conc. HNO3 [Given: Atomic mass of H = 1, C = 12, N = 14, 0= 16, S = 32, Cl = 35] Q (major product)
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Given the major product formed when each of the following amines is treated exhaustively with methyI iodide and then heated with Ag2O.Explain your reasoning. Ph N.
-
Given subspaces H and K of a vector space V, the sum of H and K, written as H + K, is the set of all vectors in V that can be written as the sum of two vectors, one in H and the other in K; that is,...
-
The mass density of water vapour at 327.6 atm and 776.4 K is 133.2 kg m-3. Given that for water Tc = 647.4 K, Pc = 218.3 atm, a = 5.464 dm6 atm mol-2, b= 0.03049 dm3 mol-1, and M= 18.02 g mol-1,...
-
One popular activity that tourists participate in when they visit Alaska is panning for gold. A gift shop by the panning center sells blocks of clay. The packaging on the clay claims that one in five...
-
The text points out that asymmetric information can have deleterious effects on market outcomes. a. Explain how asymmetric information about a hidden action or a hidden characteristic can lead to...
-
Although the CPA Canada Handbook standards provide general guidance, the application of these standards is dependent upon particular circumstances. In practice, a professional accountant may...
-
4. Conley owned video poker machines. They are outlawed in Pennsylvania, but he placed them in bars and clubs. He used profits from the machines to buy more machines. Is he guilty of money laundering?
-
The following were selected from among the transactions completed by Sorento Co. during the current year. Sorento Co. sells and installs home and business security systems. Jan. 5. Loaned $17,500...
-
I will rate the tutor HELPFUL (thumbs up) if all task/instructions are followed ++ Pleas3 share the l!nk for the g00gle sheets or excel after. THANK YOU YOU SO MUCH!. below: The trial balance of Di...
-
The previous year was Amys first year of operating the bookstore. Amy and Ken elected to carry forward a $4,752 net operating loss from the first year of business into 2015. (Note: Net operating...
-
Consider the data shown below for Vicuna, a country that produces only two products oranges and shirts. Year 2013 2015 Oranges Produced 1,800 2,000 Price of Oranges $0.90 1.00 Shirts Produced 110...
-
The revenue function R(x) and the cost function C(x) for a particular product are given. These functions are valid only for the specified range of values. Find the number of units that must be...
-
How is it that anti - accident can serve too slow the damage caused by free radicals?
-
You are measuring the length of a string using a a meter stick. Your actual measurement of the string is 58 cm 1 cm. Compare the accuracy and precision of your measurement compared to the expected...
-
Find the nominal annual rate of interest for the following investment. Principal $1177.00 Future Value $2867.12 Time Due 8 years Frequency of Conversion monthly The nominal annual rate of interest is...
-
Draw a sketch depicting the same charges: Q1, Q2 and Q3. Include the appropriate labels for the sign of each charge and place an asterisk on Q3. Draw arrows on the sketch to show: 1) The direction of...
-
To test if the variance of population 2 is higher than the variance of population 1 at a 10% level of significance, we can use the F-test for equality of variances. The null hypothesis is that the...
-
The baseball player A hits the ball from a height of 3.36 ft with an initial velocity of 34.8 ft/s. 0.14 seconds after the ball is hit, player B who is standing 15 ft away from home plate begins to...
-
Calculate the wavelength of the Balmer line of the hydrogen spectrum in which the initial n quantum number is 5 and the final n quantum number is 2.
-
A flask contains 201 mL of argon at 21oC and 738 mmHg. What is the volume of gas, corrected to STP?
-
The equilibrium constant Kc for the synthesis of methanol, CH 3 OH, CO(g) + 2H 2 (g) CH 3 OH(g) is 4.3 at 250C and 1.8 at 275C. Is this reaction endothermic or exothermic?
-
Question: Joel Curtis was two years old and his brother, Joshua, was three years old when their father left both children asleep in the rear seat of his automobile while visiting a friend. His...
-
1. YOU BE THE JUDGE WRITING PROBLEM Process cheese food slices must contain at least 51 percent natural cheese. Imitation cheese slices, by contrast, contain little or no natural cheese and consist...
-
2. Josephine Rutyna was a 60-year-old widow who suffered from high blood pressure and epilepsy. A bill collector from Collections Accounts Terminal, Inc., called her and demanded that she pay $56 she...

Study smarter with the SolutionInn App



