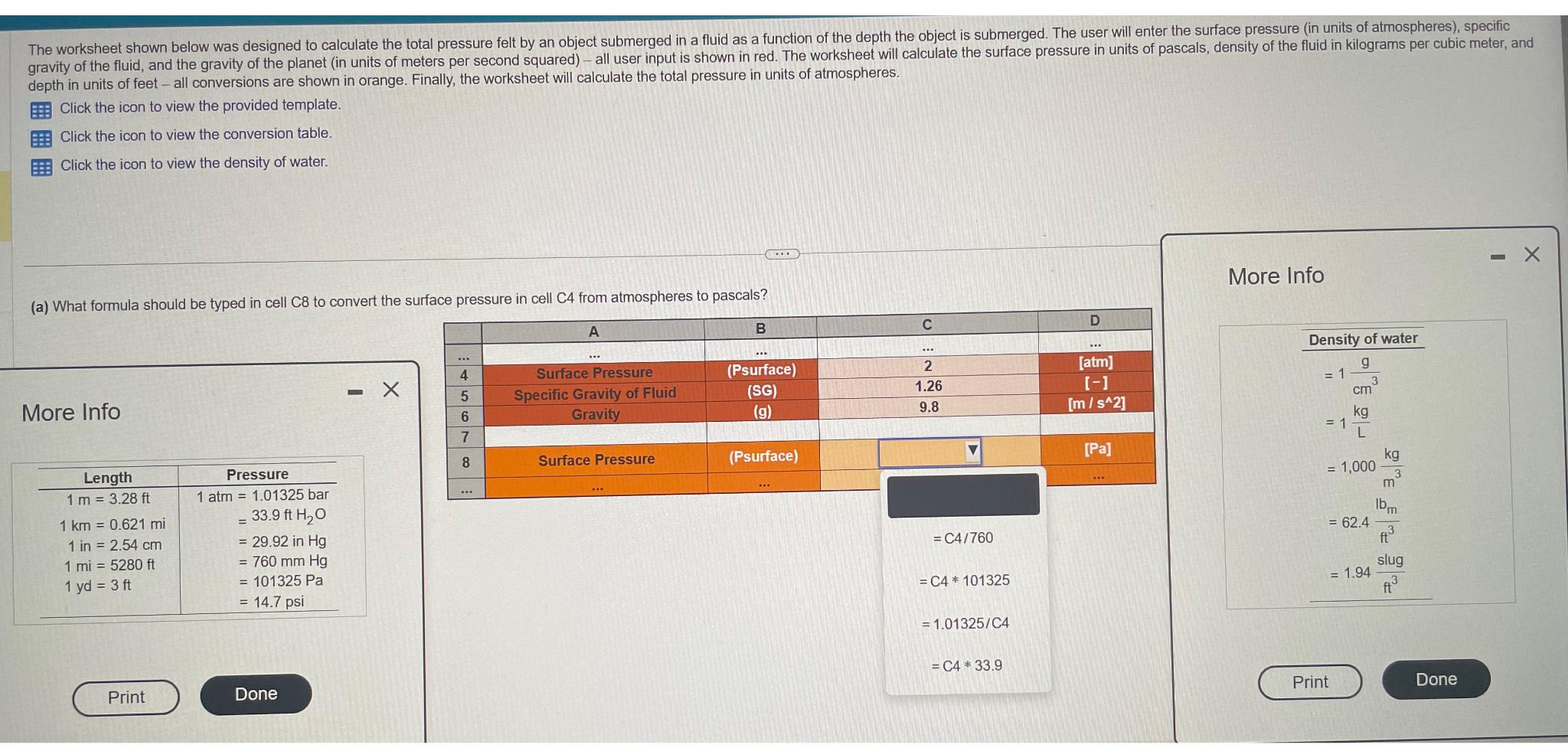
The worksheet shown below was designed to calculate the total pressure felt by an object submerged...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The worksheet shown below was designed to calculate the total pressure felt by an object submerged in a fluid as a function of the depth the object is submerged. The user will enter the surface pressure (in units of atmospheres), specific gravity of the fluid, and the gravity of the planet (in units of meters per second squared) - all user input is shown in red. The worksheet will calculate the surface pressure in units of pascals, density of the fluid in kilograms per cubic meter, and depth in units of feet - all conversions are shown in orange. Finally, the worksheet will calculate the total pressure in units of atmospheres. Click the icon to view the provided template. Click the icon to view the conversion table. Click the icon to view the density of water. (a) What formula should be typed in cell C8 to convert the surface pressure in cell C4 from atmospheres to pascals? A More Info Length 1 m = 3.28 ft 1 km = 0.621 mi 1 in = 2.54 cm 1 mi = 5280 ft 1 yd = 3 ft Print Pressure 1 atm 1.01325 bar 33.9 ft HO = 29.92 in Hg = 760 mm Hg = 101325 Pa = 14.7 psi = Done 4 5678 www www Surface Pressure Specific Gravity of Fluid Gravity Surface Pressure B (Psurface) (SG) (g) (Psurface) www C ... 2 1.26 9.8 =C4/760 = C4 * 101325 = 1.01325/C4 = C4 33.9 D [atm] [-] [m/s^2] [Pa] More Info Density of water g 3 = 1 = 1 cm kg = 1,000 = 62.4 Print = 1.94 kg 3 m lbm ft slug ft Done The worksheet shown below was designed to calculate the total pressure felt by an object submerged in a fluid as a function of the depth the object is submerged. The user will enter the surface pressure (in units of atmospheres), specific gravity of the fluid, and the gravity of the planet (in units of meters per second squared) - all user input is shown in red. The worksheet will calculate the surface pressure in units of pascals, density of the fluid in kilograms per cubic meter, and depth in units of feet - all conversions are shown in orange. Finally, the worksheet will calculate the total pressure in units of atmospheres. Click the icon to view the provided template. Click the icon to view the conversion table. Click the icon to view the density of water. (a) What formula should be typed in cell C8 to convert the surface pressure in cell C4 from atmospheres to pascals? A More Info Length 1 m = 3.28 ft 1 km = 0.621 mi 1 in = 2.54 cm 1 mi = 5280 ft 1 yd = 3 ft Print Pressure 1 atm 1.01325 bar 33.9 ft HO = 29.92 in Hg = 760 mm Hg = 101325 Pa = 14.7 psi = Done 4 5678 www www Surface Pressure Specific Gravity of Fluid Gravity Surface Pressure B (Psurface) (SG) (g) (Psurface) www C ... 2 1.26 9.8 =C4/760 = C4 * 101325 = 1.01325/C4 = C4 33.9 D [atm] [-] [m/s^2] [Pa] More Info Density of water g 3 = 1 = 1 cm kg = 1,000 = 62.4 Print = 1.94 kg 3 m lbm ft slug ft Done
Expert Answer:
Answer rating: 100% (QA)
The image shows a worksheet with different cells containing valu... View the full answer

Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
A projectile fired from the point (0, 0) at an angle to the positive x-axis has a trajectory given by where x = horizontal distance in meters y = height in meters v = initial muzzle velocity in...
-
answer the question clearly You are building a flight-control system for which a convincing safety case must be made. Would you assign the tasks of safety requirements engineering, test case...
-
Provide the appropriate statute for your answer (format style - IRAC - Issue, Rule, Application and Conclusion.) what are your thought? Problem Scenario- Earnest is married to Janice. Earnest and...
-
Knowing that = 60, determine the reaction (a) at B, (b) at C.
-
Choosing any set of numbers to represent utils, draw a total utility curve that shows your increasing enjoymentthen decreasing enjoymentas you consume more and more chocolate-covered donuts. Draw the...
-
Let \(Z\) be an \(\mathbb{R}\)-valued semi-martingale such that \(Z\) and \(Z_{-}\) do not vanish. Prove that \[ \mathcal{L}(Z)_{t}=\ln \left(\left|\frac{Z_{t}}{Z_{0}} ight| ight)+\int_{0}^{t}...
-
1. What are some factors that Alan should consider when determining whether or not to offer benefits to part-time workers? 2. Do you think the firm should offer benefits to part-time workers? If yes,...
-
Identify the effects of the following transactions in the table below: Balance Sheet Income Statement Cash Transaction + Asset Noncash Assets Contrib. Earned Net Liabilities + + - Revenues Expenses =...
-
When possible, compute the truth values of the simple components in the following compound propositions. If no truth value can be computed, write a question mark (?) under the letter or letters with...
-
Thesis statement on PA SB240 An Act amending the act of July 19, 1979 (P.L.130, No.48), known as the Health Care Facilities Act, providing for hospital patient protection.
-
for the following code, what is the total LOC value?? void sort (int *a, int n) { int i, j, t, x,y; } if (n < 2) return; for (i=0; i
-
ABC Corporation, a U.S. company, sold equipment to a French company for Euro 100 million. Payment is due in 90 days. Answer the following questions (24-26) using the information below: Current spot...
-
Advance Auto Parts is evaluating a project with the following cash flows: Year 0 1 2 Cash Flow -$100,000 215,000 -115,500 If the company uses an interest rate of 7.5%, which one of the following...
-
What does the program below print if the input user entered was: 15 -3 0 Question 2. Can you tell what the program does? Write one sentence explaining what the function does, in plain English....
-
. So far, we have seem how to solve various partial differential equations through separation of variables. It is not difficult to extend this to inhomogeneous equations. Consider, for the equation...
-
A mass of 500 g of gaseous ammonia (polar gas) is contained in a vessel of 30,000 cm3 volume and immersed in a constant-temperature bath at 65C. Calculate the P of the gas by (a) the ideal gas EOS...
-
The Ranch 888 Noodle Company sells two types of dried noodles:ramen, at $6.50 per box, and chow fun, at $7.70 per box. So farthis year, the company has sold a total of 110,096 boxes ofnoodles,...
-
The gaseous reaction A B has a unimolecular reaction rate constant of 0.0015 min1 at 80F. This reaction is to be carried out in parallel tubes 10 ft long and 1 in. inside diameter, under a pressure...
-
Review Figure 12-13. Use this figure to write a few sentences (or at least draw an analogy) explaining why, when you strike the head of a safety match slowly on its pumice with little pressure, it...
-
An Algorithm for Management of Change (MoC). List three things that would not have occurred if the MoC algorithm had been followed.
-
The following information is available for McKee Corporation for 2015. 1. Excess of tax depreciation over book depreciation, 40,000. This 40,000 difference will reverse equally over the years...
-
In 2015, Amirante Corporation had pretax financial income of $168,000 and taxable income of $120,000. The difference is due to the use of different depreciation methods for tax and accounting...
-
Brennan Corporation began 2015 with a $90,000 balance in the Deferred Tax Liability account. At the end of 2015, the related cumulative temporary difference amounts to $350,000, and it will reverse...

Study smarter with the SolutionInn App


